Biologics Manufacturing Market Share, Size, Trends, Industry Analysis Report, 2026 - 2034
REPORT DETAILS
REPORT DETAILS
ABOUT THIS REPORT
Biologics Manufacturing Market Summary
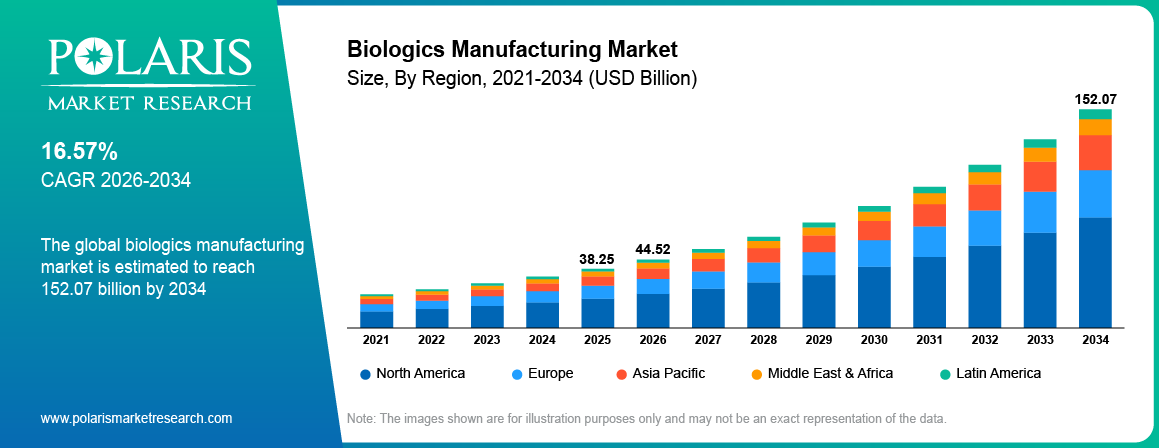
The global biologics manufacturing market is estimated around USD 38.25 Billion in 2025, with consistent growth anticipated during 2026–2034. This growth is driven by increasing demand for biologics in drug development, rising prevalence of chronic diseases, and expanding adoption of targeted therapies across oncology, autoimmune disorders, and infectious diseases. The market is projected to grow at a CAGR of 16.57% during the forecast period.
Key Takeaways:
- Monoclonal antibodies segment dominated with 47.20% share in 2025 due to strong demand for targeted therapies across multiple therapeutic areas.
- Single-use systems segment led with a revenue share of 32.00% in 2025 owing to increasing adoption of flexible and cost-efficient manufacturing solutions.
- Vaccines segment is projected to grow rapidly at a CAGR of 15.61%, due to rising focus on preventive healthcare and immunization programs.
- North America dominated the market capturing 45.40% share, due to strong R&D investments and advanced biopharmaceutical infrastructure.
- Key players operating in the market include AbbVie Inc., Amgen Inc., Boehringer Ingelheim International GmbH, Bristol-Myers Squibb Company, Catalent, Inc., Celltrion Inc., Eli Lilly and Company, F. Hoffmann-La Roche Ltd., FUJIFILM Diosynth Biotechnologies, Johnson & Johnson, Lonza Group AG, Novartis AG, Pfizer Inc., Samsung Biologics Co., Ltd., and WuXi Biologics (Cayman) Inc.
Industry Dynamics
- Rising demand for targeted therapies increases need for biologics production.
- Increasing pharmaceutical investments support expansion of manufacturing capacity.
- High capital investment and regulatory complexity limit market entry.
- Growth in biosimilars and emerging markets creates new opportunities.
Market Statistics
- 2025 Market Size: USD 38.25 Billion
- 2034 Projected Market Size: USD 152.07 Billion
- CAGR (2026-2034): 16.57%
- North America: Largest market in 2025
What is Biologics Manufacturing?
Biologics manufacturing represents a critical segment of the pharmaceutical industry, focused on producing complex biological molecules through advanced bioprocessing systems. Biologics manufacturing involves the use of living cells and microorganisms to develop therapies such as monoclonal antibodies, vaccines, and recombinant proteins. The process of biologics manufacturing involves several steps including cell culture, fermentation, purification, and quality control, among others, which necessitate unique equipment and skilled personnel.
Rising demand for biologics manufacturing is attribute to the growing share of biologics in drug development and approvals. Biologics make up a large number of drug approvals worldwide, representing the change in the direction of drugs being developed. This trend is owing to the change in focus from general medication production to specialized medications. Increase in the number of chronic diseases and the requirement for effective medication have led to the biologics production process.
To Understand More About this Research: Download Sample Report
Biopharmaceutical and biotech firms are upgrading their manufacturing capacity as a response to growing demand for biologics and ensuring stable manufacturing processes. Cell culture techniques and process engineering are making significant contributions to enhanced production efficiency and scalable manufacturing operations. In addition, contract manufacturers play a key role in the manufacture of biologics through providing flexible and economical manufacturing options.
Drivers & Opportunities
Increasing Demand for Targeted Therapies Fueling the Growth of Biologics Manufacturing: The growing demand for targeted therapies like monoclonal antibodies and gene therapies is increasing the demand for biologics. In May 2025, Invivyd launched a pipeline initiative that aimed at developing monoclonal antibodies to treat infectious diseases including measles in addition to COVD-19, RSV, and influenza.This was through use of predictive modeling coupled with engineering antibodies to be resistant to viruses’ mutations. These developments emphasizes the growing reliance on precision biologics in oncology and infectious disease treatment.
Increasing pharmaceutical investments support expansion of Biologics Production Capacity: Pharmaceutical firms are investing more in biologics product lines and manufacturing processes. In April 2026, AbbVie announced investment of USD 1.4 billion towards setting up an industrial facility in North Carolina with the focus on developing injectable drugs for immunology, oncology, and neurology domains.The company aimed to utilize cutting-edge manufacturing technology to enhance efficiency and capability in the manufacturing process.
Restraints & Challenges
High Capital Investment and Regulatory Complexity Limit Market Entry: Large investment in terms of capital expenditure (CAPEX) is needed to set up facilities for biologics manufacturing and the need for complex machinery and compliance systems makes things even harder. Complex regulation systems concerning quality, safety, and validation contribute to make the business difficult. These factors hinders the market growth for new entrants and extend timeline for launching the product in the market.
Opportunity
Emerging Markets and Biosimilars Create Strong Biologics Opportunities: The development of efficient ecosystems for manufacturing operations in developing regions like the Asia Pacific is driving the manufacture of biologics. Nations from this region are building their capabilities and harmonizing their regulations to host biopharmaceutical manufacturing facilities. There is an increase in demand for biosimilars that are making biological medicines more accessible at low costs and generating additional revenues for manufacturers.

Segmental Insights
This report provides a detailed assessment of the biologics manufacturing market across product and technology to identify leading segments and emerging growth areas.
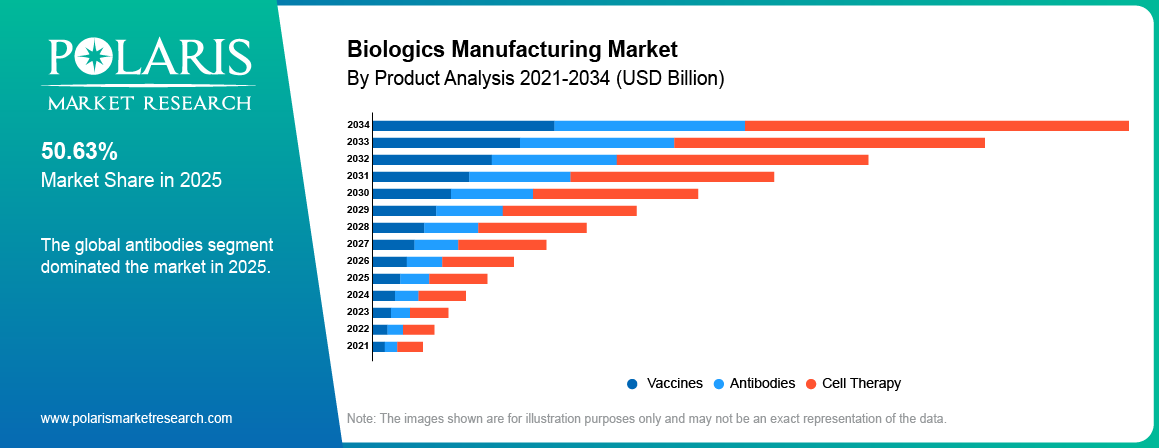
By Product
-
Monoclonal Antibodies
The monoclonal antibodies segment accounted for the largest share accounting for 47.20% in 2025 due to strong demand for targeted therapies in oncology and autoimmune diseases. Rising approvals along with growing pipelines are making it possible for companies to scale up their productions. Ongoing investments in big-scale productions help to promote the monoclonal antibodies market and biologics segmentation.
-
Vaccines
The vaccines segment is projected to grow at a notable pace at a CAGR of 15.61%, during the forecast period driven by rising demand for preventive healthcare and immunization programs. Expansion of global vaccination initiatives and development of next-generation vaccines support manufacturing growth.
By Technology
-
Single-Use Systems
Single-use systems segment dominated the market with 32.00% in 2025 driven by increasing adoption of flexible and cost-efficient manufacturing solutions. These systems reduce contamination risk and eliminate the need for extensive cleaning processes, which improves operational efficiency.
-
Upstream & Downstream Processing
Upstream and downstream processing segment is projected to grow rapidly with a CAGR of 16.12% owing to the constant innovations in cell cultivation and product purification. Upstream processing deals with cell cultivation and optimization, while downstream processing ensures purity of the product.
Regional Analysis
North America Biologics Manufacturing Market Assessment
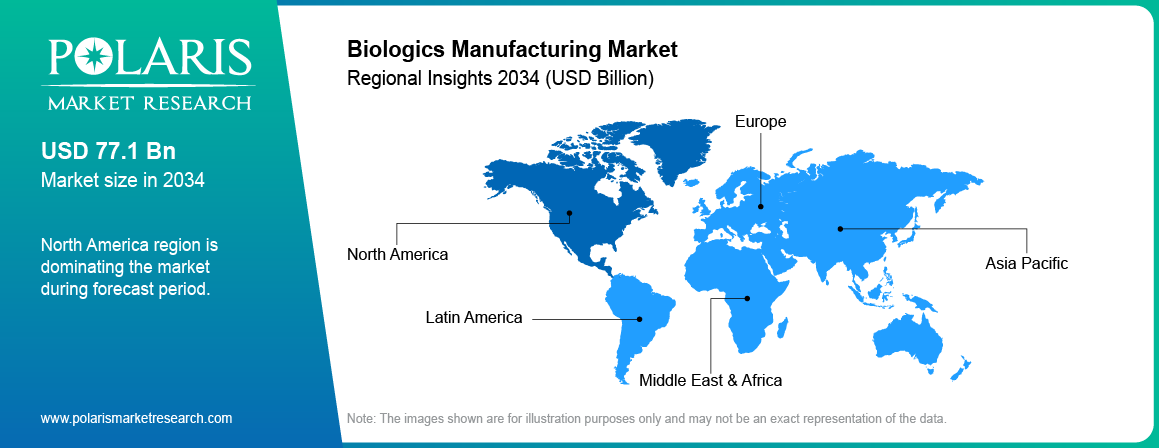
North America is leading the market share capturing 45.40% in 2025, due to substantial investments in research & development and advanced manufacturing capacity. The region has an established biopharmaceutical chain and attracts massive levels of investment. As indicated by the International Trade Administration, majority-owned firms of foreign origin recorded USD 25.71 billion in biopharmaceutical exports in the US and invested approximately USD 25.85 billion in R&D in 2022.Moreover, foreign direct investment (FDI) in the pharmaceutical industry was valued at USD 503.4 billion in 2023. This strong financial and research base supports large-scale biologics production and innovation across the region.
Asia Pacific Biologics Manufacturing Market Insights
Asia Pacific is projected to grow at a fast pace at a CAGR of 19.30%, during the forecast period, owing to rising levels of outsourcing operations, low-cost production, and growing capacity for CDMOs within nations such as India and China. The region is home to international biopharmaceutical manufacturers searching for more effective and scalable manufacturing facilities. For instance, Shilpa Medicare launched its unique CDMO model in India at the DCAT 2025 conference held in March 2025.Growth in the manufacturing base and access to a competent labor force further enhance the region’s standing in biologics manufacturing.
Europe Biologics Manufacturing Market Overview
In 2025, Europe was the second largest market valued at 28.40% in biologics manufacturing due to robust regulatory environment and efforts towards advancing the use of biologicals in therapy. The region experiences high penetration of biologics in treating rare diseases and personalized medicine. There are already biopharmaceutical companies, coupled with collaboration between institutes and manufacturers, which is driving innovation in biologics manufacture. Efforts by government towards fostering local capacity in biologics manufacture contribute to the growth of the market.
Competitive Landscape & Key Players
Biologics manufacture market is characterized by moderate consolidation whereby large biopharmaceutical firms and contract development and manufacturing organization compete to meet increasing demands for biological products. In such a market, players engage in activities geared towards enhancing their manufacturing capacity and efficiency to cope with growing production needs.
The key players in the market include AbbVie Inc., Amgen Inc., Boehringer Ingelheim International GmbH, Bristol-Myers Squibb Company, Catalent, Inc., Celltrion Inc., Eli Lilly and Company, F. Hoffmann-La Roche Ltd., FUJIFILM Diosynth Biotechnologies, Johnson & Johnson, Lonza Group AG, Novartis AG, Pfizer Inc., Samsung Biologics Co., Ltd., and WuXi Biologics (Cayman) Inc.
Premium Insights
-
Regulatory Framework
The regulatory framework for the biologics manufacturing market focuses on product quality, process consistency, and compliance with GMP standards. The U.S. Food and Drug Administration, the European Medicines Agency, and other authorities have put in place strict policies on how biologics manufacturing should be done, validated, and monitored for quality. Manufacturers are mandated to guarantee consistency and traceability throughout the manufacturing process and during regulatory compliance protocols.
-
Key Risks Associated with Biologics Manufacturing Market
There are several risks associated with the high cost of producing biologics and the complicated nature of the manufacturing process. Biological variability also pose challenges to product consistency and yield. Other risks include regulatory compliance, large-scale manufacturing capacities, and disruptions in the supply chain.
-
Strategic Insights
Continuous manufacturing processes for biologics are the way to go in the future. AI biologics are making manufacturing processes more efficient through process optimizations and better yield and predictive maintenance. The use of modular biologics manufacturing is also picking up owing to their flexible nature and ease of setup.
Key Players
- AbbVie Inc.
- Amgen Inc.
- Boehringer Ingelheim International GmbH
- Bristol-Myers Squibb Company
- Catalent, Inc.
- Celltrion Inc.
- Eli Lilly and Company
- F. Hoffmann-La Roche Ltd.
- FUJIFILM Diosynth Biotechnologies
- Johnson & Johnson
- Lonza Group AG
- Novartis AG
- Pfizer Inc.
- Samsung Biologics Co., Ltd.
- WuXi Biologics (Cayman) Inc.
Industry Developments
- January 2026: Wuxi Biologics launched the PATROLab digital twin system to generate simulations for real-time manufacturing processes. [source: wuxibiologics.com]
- October 2025: Samsung Biologics launched ExellenS manufacturing platform for harmonizing the manufacturing processes and equipment within the organization. [source: samsungbiologics.com]
Biologics Manufacturing Market Segmentation
By Product Outlook (Revenue, USD Billion, 2021-2034)
- Vaccines
- Antibodies
- Cell Therapy
By Technology Outlook (Revenue, USD Billion, 2021-2034)
- Single-Use
- Upstream
- Downstream
By Regional Outlook (Revenue, USD Billion, 2021-2034)
- North America
- US
- Canada
- Europe
- Germany
- France
- UK
- Italy
- Spain
- Netherlands
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Malaysia
- South Korea
- Indonesia
- Australia
- Vietnam
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Israel
- South Africa
- Rest of Middle East & Africa
- Latin America
- Mexico
- Brazil
- Argentina
- Rest of Latin America
Biologics Manufacturing Market Report Scope
|
Report Attributes |
Details |
|
Market Size in 2025 |
USD 38.25 Billion |
|
Market Size in 2026 |
USD 44.52 Billion |
|
Revenue Forecast by 2034 |
USD 152.07 Billion |
|
CAGR |
16.57% from 2026 to 2034 |
|
Base Year |
2025 |
|
Historical Data |
2021–2024 |
|
Forecast Period |
2026–2034 |
|
Quantitative Units |
Revenue in USD Billion and CAGR from 2026 to 2034 |
|
Report Coverage |
Revenue Forecast, Competitive Landscape, Growth Factors, and Industry Trends |
|
Segments Covered |
|
|
Regional Scope |
|
|
Competitive Landscape |
|
|
Report Format |
|
|
Customization |
Report customization as per your requirements with respect to countries, regions, and segmentation. |
FAQ's
The global market size was valued at USD 38.25 Billion in 2025 and is projected to grow to USD 152.07 Billion by 2034.
North America held a leading position with a revenue share of 45.40% owing to substantial investments in research and development along with advanced manufacturing capabilities.
Major applications include monoclonal antibodies, vaccines, recombinant proteins, and advanced therapeutic products.
A few of the key players in the market are AbbVie Inc., Amgen Inc., Boehringer Ingelheim International GmbH, Bristol-Myers Squibb Company, Catalent, Inc., Celltrion Inc., Eli Lilly and Company, F. Hoffmann-La Roche Ltd., FUJIFILM Diosynth Biotechnologies, Johnson & Johnson, Lonza Group AG, Novartis AG, Pfizer Inc., Samsung Biologics Co., Ltd., and WuXi Biologics (Cayman) Inc.
Growth is driven by increasing biologics approvals, rising demand for targeted therapies, and expanding pharmaceutical investments.
High capital expenditures and stringent regulations make operations difficult.
Pharmaceutical companies, biotechnology companies, and contract manufacturing organizations drive the demand.
The market outlook remains strong due to expansion of advanced therapies and increasing global demand for biologics.
Rising demand for affordable biological medicines boosts biosimilars manufacturing.
Download Sample Report of Biologics Manufacturing Market
Please fill out the form to request a customized copy of the research report.
