What is Amniocentesis Needle Market Size?
Amniocentesis Needle Market size was valued at USD 213.01 million in 2025 and is anticipated to exhibit the CAGR of 7.3% during the forecast period. The growth is driven by gestational disorder, rising preference for non-invasive pernatel tests, and technological advancement.
Market Statistics
- 2025 Market Size: USD 213.01 Million
- 2034 Projected Market Size: USD 399.24 Million
- CAGR (2026-2034): 7.3%
- Largest Market: North America
Industry Overview
A prenatal procedure called amniocentesis is performed to identify infections and chromosomal abnormalities in fetuses. A small amount of fluid, 30 cubic centimeters, is taken from the womb for this test. This is then utilized to identify any hereditary diseases that the infants may have. The primary cause of the market's expansion is the rise in genetic disease frequency on a global scale. Healthcare professionals are recommending amniocentesis procedures more frequently, with a higher success rate. This is likely due to increased awareness of congenital diseases, including Down's syndrome. Additionally, there is a growing population of pregnant individuals and increased investment in healthcare research and development. These factors are expected to drive sales of amniocentesis needles.
In March 2021, The BD Chorionic Villus Sampling (CVS) System, a novel amniocentesis needle introduced by Becton Dickinson and Company, has a revolutionary ergonomic handle design that reduces hand fatigue and increases accuracy throughout operations.
With increasing pregnancy rates and the widespread use of diagnostic tests in developed countries, the amniocentesis market is expected to grow in the coming years. Furthermore, a high occurrence of congenital abnormalities and a rise in middle-aged mothers during the projected period will also be the factors to fuel the market.
To Understand More About this Research: Request a Free Sample Report
Furthermore, technological advancements in healthcare, specifically within prenatal diagnostics, play a pivotal role. Ongoing enhancements in the design of amniocentesis needles and diagnostic techniques have significantly improved accuracy, safety, and patient comfort. These improvements concentrate on optimizing needle structure, minimizing size, and refining procedural precision, ultimately ensuring a safer and more comfortable experience during amniocentesis procedures. Additionally, advancements in genetic testing and molecular diagnostics have bolstered the thorough examination of samples acquired through amniocentesis. This progress markedly enhances the precision in identifying various genetic abnormalities, thereby contributing to an overall improved experience.
Amniocentesis Needle Market Dynamics
Market Drivers
Rising Occurrence of Gestational Diabetes bolstering the growth of the Amniocentesis Needle market industry.
The global surge in diabetes cases has led to numerous health complications for many individuals. The elevated prevalence of gestational diabetes has consequently increased the demand for amniocentesis, particularly in pregnant women with diabetes who experience a heightened accumulation of amniotic fluid. This condition poses risks to both maternal and fetal health, driving the need for amniocentesis needles to alleviate complications by draining excess amniotic fluid and addressing the health concerns of pregnant women.
Market Restraints
Rising Preference for Non-Invasive Prenatal Tests (NIPTs) is likely to hamper the growth of the market.
The rising popularity of minimally invasive procedures is anticipated to continue growing in the foreseeable future, driven by research advancements and increased accessibility to non-invasive procedures. This trend, however, poses a potential challenge for the amniocentesis needle market revenue, as these procedures often do not necessitate the use of needles. Additionally, Non-Invasive Prenatal Tests (NIPTs) offer advantages such as reduced infection risk and cross-contamination, further impacting the demand for amniocentesis needles. Companies are responding to this shift by introducing new non-invasive prenatal tests to meet the demand generated by advancements in minimally invasive technologies, potentially posing a threat to the growth of the amniocentesis needle market forecast in the near future. For instance, In April 2021, ARCEDI Biotech ApS introduced EVITA TEST COMPLETE, a prenatal genetic blood test capable of isolating and identifying fetal cells in maternal blood, providing comprehensive information on 22 chromosome pairs and sex chromosomes as early as the 12th week of pregnancy.
Key Trends in Amniocentesis Needle
|
Trend |
Description |
Key Drivers |
Examples from Practice |
|
Enhanced echogenic coatings |
Surface modifications (laser etching, polymer coatings, dimpling) improve ultrasound visibility of needle tip and shaft during real‑time guidance. |
Reducing failed insertions and complications by enabling precise visualization in free‑hand or needle‑guide techniques for amniocentesis. |
Systematic review compares echogenic enhancements (laser, polymer, dimpled), finding significant visibility improvements across techniques; BD's 2024 echogenic tip boosts visibility by 42%. |
|
Thinner gauge and atraumatic designs |
Shift to finer gauges (22G or thinner) with tapered, pencil‑point or beveled atraumatic tips to lower maternal/fetal trauma and post‑procedure discomfort. |
Single‑insertion success in high‑risk pregnancies, minimizing miscarriage risk (0.1–0.3%) and amniotic fluid leakage. |
Medtronic's 2023 next‑gen needle features thinner gauge and atraumatic tip for reduced discomfort/risk; innovations emphasize ultra‑fine echogenic designs. |
|
Fiber‑optic needle tip tracking |
Integrated fiber‑optic shape sensing provides real‑time 3D needle trajectory visualization during insertion into ex vivo/in vivo models. |
Overcoming ultrasound limitations in deep tissue or obese patients, enabling safer transplacental access if needed. |
2022 study demonstrates fiber‑optic sensing for intraoperative amniocentesis needle tracking in placenta, enhancing precision beyond standard echogenic tech. |
|
Antimicrobial peptide (AMP) coatings |
Immobilization of AMPs via physico‑chemical coatings on needle surfaces to prevent bacterial infections without promoting resistance. |
Reducing procedure‑related infections in invasive prenatal diagnostics, especially in outpatient settings. |
2021–2025 reviews detail AMP coatings on medical devices like needles, showing broad‑spectrum activity and clinical promise for infection control. |
|
Continuous ultrasound guidance protocols |
Standardized free‑hand or needle‑guide techniques with constant visualization, limiting to 1–2 punctures to avoid complications. |
Operator training for <3 punctures, using 22G spinal needles with stilette and vacuum aspiration. |
Cochrane review (2012, cited 29x) and ISUOG guidelines stress continuous guidance reduces risks; abandon after 2 failed pricks. |
|
AI‑powered ultrasound integration |
AI enhances real‑time needle tracking and anatomy recognition during amniocentesis for automated risk assessment. |
Improving accuracy in needle placement, reducing variability across operators in prenatal diagnostics. |
Industry reports note AI‑ultrasound live tracking limits adverse outcomes; design aids predict needle‑tissue interactions. |
Report Segmentation
The market is primarily segmented based on type, procedure, end-use, and region.
|
By Type |
By Procedure |
By End-Use |
By Region |
|
|
|
|
To Understand the Scope of this Report: Speak to Analyst
Amniocentesis Needle Market Segmental Analysis
By Type Analysis
The segment of needles ranging from 100 to 150 mm secured the largest market share in 2025, attributed to their versatility and applicability across various medical procedures, including drug administration, vaccinations, and blood sampling. These needles strike a balance between precision and patient comfort, making them a preferred choice in the medical field. Notably, they are frequently employed for females with a normal body mass index (BMI) and are commonly administered to pregnant women with a higher BMI. For example, in the UK, where a substantial obstetric challenge stems from a high obesity rate affecting 21.3% of pregnant women, with only 47.3% having a normal BMI, the usage of this needle is anticipated to rise in obstetric practice.
The segment of needles smaller than 100 mm exhibits the fastest CAGR throughout the forecast period. In the context of early pregnancies, there is a growing preference for shorter needles, especially when dealing with a smaller amniotic sac where maneuverability is crucial. These shorter needles align with the increasing demand for minimally invasive procedures, minimizing patient discomfort and tissue trauma. Ongoing advancements in needle design, including atraumatic tips and compatibility with ultrasound guidance, contribute to enhancing the safety and accuracy of early-trimester procedures, further fostering the adoption of smaller needles. Moreover, the medical practitioners' inclination towards smaller needle sizes, driven by improved maneuverability and reduced risk of complications, is a significant factor contributing to the heightened adoption and anticipated rapid expansion of this specific segment in the market.
By Procedure Analysis
The amniocentesis procedures segment, dominated the revenue share in 2025. This growth is primarily attributed to the significance of amniocentesis as a crucial prenatal diagnostic tool, providing valuable insights into fetal health, genetic conditions, and chromosomal abnormalities essential for expectant parents and healthcare providers. Recent years have witnessed advancements in amniocentesis techniques, including non-invasive prenatal testing (NIPT) and sophisticated genetic testing technologies, expanding its scope and enhancing accuracy. Notably, the increasing adoption of NIPT offers highly precise genetic information while avoiding the invasiveness associated with alternative procedures. The rise in maternal age and heightened awareness of genetic disorders have further propelled the demand for such prenatal assessments.
The fetal blood transfusion segment is experiencing a significant CAGR over the forecast period. Progress in fetal medicine and perinatal care has led to a heightened recognition of the effectiveness of fetal blood transfusion procedures in addressing specific fetal conditions. Additionally, advancements in needle technology, characterized by smaller gauges and atraumatic tips, are enhancing the safety and feasibility of fetal blood transfusions, consequently fostering their increased adoption. Moreover, the escalating incidences of fetal blood disorders, such as hemolytic disease, necessitating specialized interventions like intrauterine transfusions, have played a pivotal role in the expanded utilization of fetal blood transfusion procedures within this particular medical application.
Amniocentesis Needle Market Regional Insights
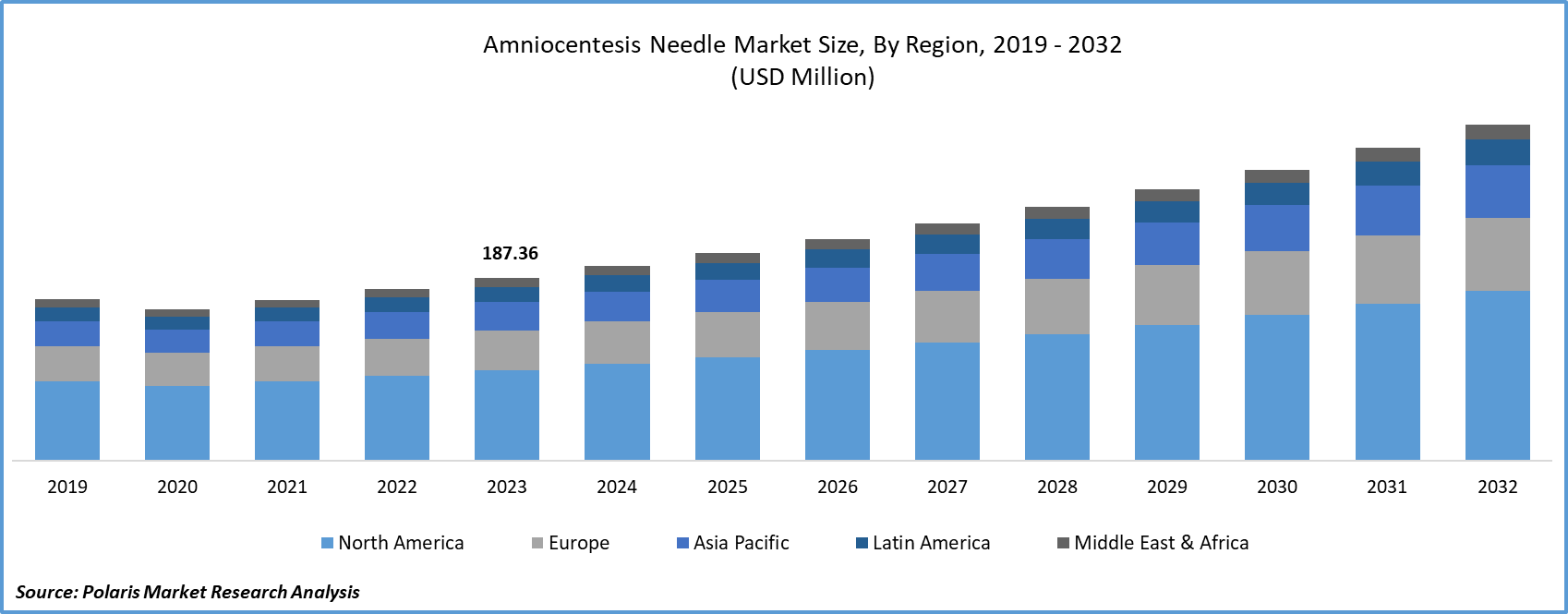
The North American region dominated the global market with the largest market share in 2025
The prominence of this is credited to a robust healthcare infrastructure, sophisticated prenatal care facilities, and a heightened awareness regarding the significance of prenatal testing among expectant parents. A growing number of couples in the U.S. and Canada are choosing prenatal screening to assess the health of their unborn babies and detect genetic conditions and chromosomal abnormalities. The presence of advanced diagnostic technologies and innovative genetic testing methods within healthcare facilities in North America has significantly bolstered the region's leading position in this domain.
The U.S. held the largest market share in North America. This can be attributed to the robust healthcare infrastructure and extensive research and development endeavors in the medical sector, resulting in technological advancements in prenatal diagnostics. The heightened demand for prenatal testing procedures, including amniocentesis, in the U.S. is driven by factors such as a higher prevalence of genetic disorders, an aging population, favorable reimbursement policies, and increased awareness among healthcare professionals and patients about the benefits of prenatal testing. These factors collectively position the U.S. as an ideal market for products like amniocentesis needles and other prenatal testing solutions when compared to other countries in North America.
Asia Pacific region is propelled to be the fastest growing region in the amniocentesis market by a rising awareness of prenatal care and the significance of genetic screening. Countries with substantial populations, such as China and India, are experiencing an escalating demand for advanced prenatal diagnostics. The Asian nations have witnessed a surge in the number of pregnant women seeking comprehensive prenatal care. Additionally, ongoing technological advancements in the region, coupled with the emergence of telemedicine and digital healthcare solutions, are extending the accessibility of prenatal testing services.
India held the largest market share in the Asia Pacific region. This can be attributed to its substantial population and a growing awareness of prenatal health and genetic concerns. The increased recognition of these factors has generated a heightened demand for advanced prenatal diagnostic techniques such as amniocentesis. This increased awareness has particularly underscored the significance of early and accurate diagnosis, especially within a significant population, thereby contributing significantly to India's prominence in the Asia Pacific region.
Competitive Landscape
The Amniocentesis Needle market opportunities is fragmented and is anticipated to witness competition due to several players' presence. Major service providers in the market are constantly upgrading their technologies to stay ahead of the competition and to ensure efficiency, integrity, and safety. These players focus on partnership, product upgrades, and collaboration to gain a competitive edge over their peers and capture a significant market share.
Some of the major players operating in the global market include:
- Becton Dickinson and Company
- Biopsybell Srl
- Cook Medical
- CooperSurgical
- Integra LifeSciences
- Laboratoire CCD
- Medline
- RI.MOS. Srl
- Rocket Medical
- Smith’s Medical
Recent Developments
- In March 2025, scientists in Israel are evaluating a new fetal screening approach that could serve as an alternative to conventional amniocentesis. Headed by Prof. Noam Shomron at Tel Aviv University, the method relies on a standard maternal blood draw instead of withdrawing amniotic fluid via a needle, a procedure associated with miscarriage risk. The blood-based screening can be performed as early as the 10th week of pregnancy—about four weeks sooner than typical amniocentesis—and delivers similar genetic insights into syndromes, diseases, and disorders. This advancement may represent a safer and earlier breakthrough in the field of prenatal healthcare.
- In April 2023, Rocket Pharmaceuticals declared the augmentation of its leadership team to reinforce its expanding and industry-leading portfolio of AAV and LV gene therapy assets.
- In June 2023, Becton, Dickinson, and Company concluded the transaction by selling its surgical instrumentation, laparoscopic instrumentation, and sterilization container assets to STERIS plc.
- In February 2022, CooperSurgical completed the acquisition of Cook Medical's reproductive health portfolio for a significant sum of USD 875 million. This portfolio includes a variety of medical equipment tailored for fertility, obstetrics, gynecology, and in vitro fertilization (IVF).
Report Coverage
The Amniocentesis Needle market growth report emphasizes key regions across the globe to provide a better understanding of the product to the users. Also, the report provides market insights into recent developments and trends and analyzes the technologies that are gaining traction around the globe. Furthermore, the report covers an in-depth qualitative analysis pertaining to various paradigm shifts associated with the transformation of these solutions.
The report provides a detailed analysis of the market while focusing on various key aspects such as competitive analysis, type, procedure, end-use, and futuristic growth opportunities.
Amniocentesis Needle Market Report Scope
|
Report Attributes |
Details |
|
Market size in 2025 |
USD 213.01 million |
| Market size in 2026 | USD 227.47 million |
|
Revenue forecast in 2034 |
USD 399.24 million |
|
CAGR |
7.3% from 2026 – 2034 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2034 |
|
Quantitative units |
Revenue in USD million and CAGR from 2026 to 2034 |
|
Segments covered |
By Type, By Procedure, By End-Use, By Region |
|
Regional scope |
North America, Europe, Asia Pacific, Latin America; Middle East & Africa |
|
Customization |
Report customization as per your requirements with respect to countries, regions, and segmentation. |
FAQ's
The global Amniocentesis Needle market size is expected to reach USD 399.24 million by 2034
Key players in the market are Biopsybell Srl, Cook Medical, CooperSurgical, Integra LifeSciences, Laboratoire CCD
North American contribute notably towards the global Amniocentesis Needle Market
Amniocentesis Needle Market exhibiting the CAGR of 7.3% during the forecast period
The Amniocentesis Needle Market report covering key segments are type, procedure, end-use, and region.
Research Methodology
A robust system of research, verification, and forecasting designed to ensure reliable and actionable market insights.
Polaris Market Research uses a clear and structured approach to deliver insights that clients can rely on. The process combines detailed primary and secondary research, including direct communication with industry experts. The detailed information helps build a complete picture of market trends and developments. Secondary data is gathered from credible sources such as industry reports, company filings, government source links, and trusted organization databases. It is then cross-checked through discussions with key stakeholders across the value chain. Market size and forecasts are developed using both bottom-up and top-down methods to ensure accuracy and consistency in the final results.
Project Setup
Step 1 & 2:
- We start every project by clearly understanding the client’s objective or goal, then defining the market scope, and aligning regions, segments, and timelines.
- Once the foundation is set, we collect data from all-around of sources, including company reports, government databases, and paid industry platforms.
- Our research is based on secondary data, which helps us build a strong understanding of the market across regions and industries. Then we validate this information through primary research by speaking directly with industry experts, companies, and stakeholders.
- By combining secondary and primary research, we ensure that our market insights are accurate, practical, and closely aligned with real market conditions.
Data Collection
We gather information from both public and verified sources:
Data Structuring
Step 3:
- All collected data is organized into a consistent format to ensure accurate analysis. Since inputs come from multiple sources, they are standardized and aligned before use.
- The data is segmented by product, application, and region, and mapped across a defined historical period (2020–2024). All values are converted into common units (USD Mn/Bn), and volume and pricing are aligned where required to estimate revenue.
- Any overlaps or inconsistencies are reviewed and adjusted to maintain accuracy (<5% variance threshold).
- The result is a structured dataset that allows for clear comparison across regions and supports reliable analysis and forecasting.
Structured Market Dataset, USD Mn/Bn
4. Data Structuring
Step 4: TOP-DOWN APPROACH
- We start with the overall market size at a global or macro level.
- The market is then narrowed down based on scope and industry relevance.
- We apply penetration rates and split the data by region and segment.
- This helps us estimate the market size for specific segments.
- The numbers are validated through cross-checks to ensure accuracy.
Step 5: BOTTOM-UP APPROACH
- We begin by analyzing data from leading companies in the market.
- Revenue data is collected and mapped across different segments.
- The data is then aggregated to estimate the total market size.
- To fill in any gaps, adjustments are made based on industry standards.
- Validation checks make sure that the results are correct.
5. Data Structuring
Step 6:
At Polaris Market Research, we employ a methodical forecasting strategy. This approach blends the analysis of historical data with real-time market validation. To forecast future trends with precision, we examine past patterns, pricing fluctuations, and the interplay of supply and demand. To ensure our conclusions reflect the present market landscape, we actively seek input from industry experts and key stakeholders.
To refine our predictions, we carefully consider critical elements such as market drivers and restraints, fluctuations in raw material costs, emerging technologies, and the production capabilities of various regions. Furthermore, we assess regulatory frameworks and potential policy shifts to gauge their potential impact on market expansion.
All this information is synthesized to generate precise forecasts for each segment and region. These forecasts illuminate the current state of the market and highlight forthcoming opportunities.
6. Data Structuring
Step 7:
In the final stage, we validate all our estimates using a triangulation method, where data is cross-checked from multiple reliable sources, like company data, primary interviews, and secondary research. This helps us make sure that our numbers are correct and fit with the rest of the market.
This process involves verifying data consistency across various segments and geographic areas. It also requires comparing historical trends with the assumptions support the forecast. Any discrepancies involve adjustments to ensure everything remains aligned and dependable.
Once the data is finalized, we prepare the final outputs, including market size estimates, segment-wise breakdowns, and growth metrics. These are delivered in structured formats such as tables, charts, and data files for easy analysis and use.
We collaborate closely with clients, ensuring the final products align with their requirements. This includes offering tailored adjustments, supplementary data analyses, and continuous assistance. Furthermore, we monitor market trends post-delivery, providing updates and refinements to maintain the insights' relevance as time passes.
Post-delivery, we continue to monitor market shifts, offering updates and adjustments to ensure the insights remain relevant over time.
Triangulation Framework
- Company-level data
- Primary inputs from industry participants
- Secondary benchmarks and published data
- Variance maintained within ±5-10%
- Adjustments applied to align estimates
- Segment values validated against overall market structure
Data Consistency & Integrity
- Segment totals validated to 100%
- Regional estimates aligned with global market size
- Historical trends compared against forecast outputs
- Assumptions reviewed for cross-segment and regional alignment
Final Outputs
- Market size estimates (USD Mn/Bn)
- Segment-wise distribution (%)
- Growth metrics (CAGR %)
- Structured tables and charts
- Segment-level datasets
- Excel-based data files for further analysis
Client Alignment & Support
- Deliverables are aligned with defined client requirements and scope
- Custom data cuts and segment splits are incorporated as required
- Post-delivery queries are addressed through analyst interactions
- Additional clarifications and data support are provided upon request
Client Continuity & Updates
- Market developments are tracked post-delivery to capture changes in key trends
- Updated data and revisions are provided based on new market inputs
- Additional refinements and data cuts are shared as required
- Continued analyst engagement supports evolving client requirements