What is the Current Market Size?
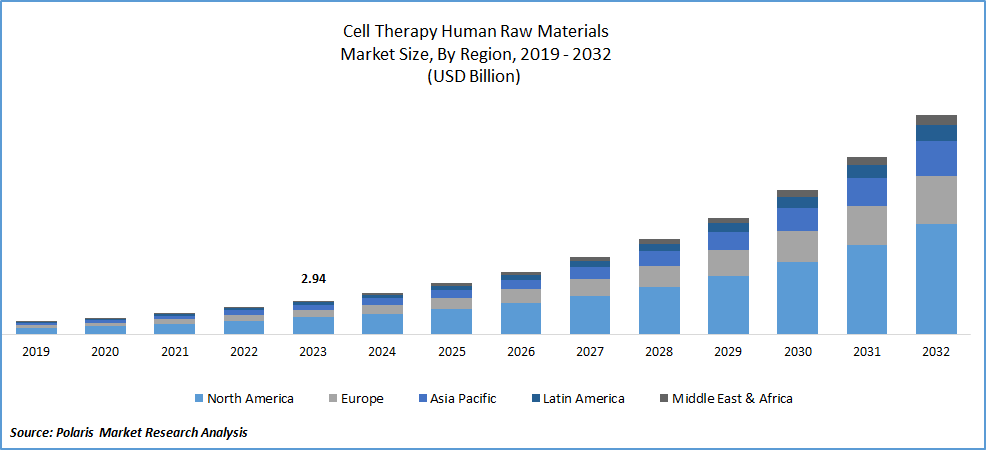
Cell therapy human raw materials market size was valued at USD 4.43 billion in 2025. The market is anticipated to grow from USD 5.43 billion in 2025 to USD 28.35 billion by 2034, exhibiting the CAGR of 22.94% during the forecast period.
Market Statistics
- 2025 Market Size: USD 4.43 billion
- 2034 Projected Market Size: USD 28.35 billion
- CAGR (2026-2034): 22.94%
- North America: Largest market in 2025
Market Introduction
The human raw materials in cell therapy is currently experiencing a surge in technological advancements, marked by the development of innovative biomaterials and sophisticated manufacturing processes. These breakthroughs serve to enhance the efficiency and scalability of cell therapy production. Also, there is a noticeable shift towards personalized medicine, placing an increased focus on tailoring cell therapies to individual patient profiles. This evolving trend underscores the imperative for a diverse range of high-quality human raw materials that are essential in supporting the manufacturing of customized cell therapies, eventually contributing to cell therapy human raw materials market growth.
Automation is gaining momentum in the manufacturing processes of cell therapy, aiming to streamline operations and enhance reproducibility. The inclination towards automated workflows in the handling of raw materials reduces the risk of contamination and ensures a consistent approach in cell therapy production.
To Understand More About this Research: Request a Free Sample Report
- For instance, in May 2023, Lonza introduced the TheraPeak T-Vivo cell culture medium, an innovative chemically defined medium designed to enhance the manufacturing of CAR T cells. Devoid of animal-origin components, this medium enhances consistency and process control, streamlining regulatory approval processes for accelerated time-to-market.
The rising incidence of chronic conditions such as cancer, diabetes, and autoimmune disorders, coupled with a growing demand for regenerative and personalized medicines, increasing research and development initiatives, and a surge in investments, are expected to increase the cell therapy human raw materials market size in the forecast period.
Industry Growth Drivers
Increasing occurrence of chronic ailments is projected to spur the product demand.
The cell therapy human raw materials market growth is greatly aided by the mounting prevalence of chronic conditions, including cancer and diabetes, stands as a significant catalyst for the cell therapy market. Cell therapies present inventive treatment avenues that address the underlying causes of these ailments, thereby amplifying the need for human raw materials.
Favorable Regulatory Landscape is expected to drive cell therapy human raw materials market growth.
Governments and regulatory entities are progressively acknowledging the promise of cell therapies and are streamlining the regulatory framework to facilitate their development and commercialization. A regulatory environment that is supportive fosters investments and expedites the expansion of the cell therapy human raw materials market size.

Industry Challenges
High development cost is likely to impede the market cell therapy human raw materials growth opportunities.
High development cost stand as a prominent factor hindering the growth of the cell therapy human raw materials market. The creation of cell therapies entails substantial research and development expenditures, encompassing costs associated with clinical trials and manufacturing processes. The substantial upfront costs may serve as a hindrance for smaller companies, constraining their engagement in the market.
Moreover, cell therapy manufacturing involves intricate processes demanding precision and adherence to stringent quality benchmarks. The intricacy of production poses challenges in scalability, impacting the cost-effectiveness of cell therapies. Thereby, hindering the cell therapy human raw materials market growth.
Critical Material Attributes for Viral Vector Production
|
Raw Material Category |
Manufacturer |
Critical Material Attributes (CMAs) |
Rationale / Testing Specified |
Supporting Documentation |
|
Transfection reagents (e.g., FectoPRO-AAV GMP) |
Sartorius (Polyplus) |
Identity, purity, potency, safety, quality; full batch traceability from synthesis to fill-finish; compliance with EudraLex Vol 4 Part II / 21 CFR Part 210. |
Ensures high transfection efficiency, batch reproducibility, no impurities impacting vector yield/safety; aseptic process simulations per Annex 1. |
Certificate of Analysis (CoA), Certificate of Origin, Certificate of Compliance, full batch record (ALCOA+ data integrity). |
|
Plasmid DNA |
Sartorius (Polyplus) |
cGMP-grade; identity, purity, high quality. |
Critical for upstream productivity; poor quality leads to low vector titers/batch failure. |
CoA confirming specs; supplier qualification. |
|
Cell culture media & components (growth factors, serum, excipients) |
Sartorius (Polyplus) |
Identity, purity, quality; animal-free preferred; full traceability. |
Prevents contamination/adventitious agents; ensures reproducibility in cell expansion/transfection. |
CoA, origin certs; risk-based QbD evaluation. |
|
Producer cells / cell banks (for unpurified bulk) |
MilliporeSigma |
Identity verification; absence of replication-competent vectors, microorganisms (bacteria, fungi, mycoplasma), adventitious viruses. |
Reduces risk of contamination/mutagenesis in vector production for cell therapy transduction. |
Rigorous testing program; master/working cell banks (MCB/WCB) qualified. |
|
Plasmids & media/serum |
MilliporeSigma |
Approved supplier sourcing; purity, identity, free of adventitious agents. |
Critical inputs must undergo testing to avoid introducing agents into process. |
Supplier qualification; CoA; broad virus assays. |
|
Final vector-related inputs |
MilliporeSigma |
Identity (gene of interest sequencing), potency (infectivity/genomic titer), sterility, endotoxin levels, aggregation, pH, osmolality. |
Ensures vector suitability for ex vivo cell therapy; links back to raw material quality. |
QC methods for unpurified bulk/final product. |
|
Cell culture media (e.g., TheraPEAK sfAAV, T-VIVO, X-VIVO) |
Lonza Bioscience |
GMP standards per USP/EP for CGT raw materials; NAO traceability; performance matching research-grade; WFI (USP/EP). |
Enables scalable, efficient vector production/transduction without animal-derived risks. |
ISO 13485 QMS; FDA-registered sites; full validation/testing docs. |
|
Freezing/lysis buffers (e.g., ProFreeze, ACK Lysing, PBS) |
Lonza Bioscience |
GMP production; safety assessments; effective removal pre-patient exposure. |
Supports cell banking/processing in vector workflows. |
Compliance certs; not for in vivo use without user validation. |
|
Nucleofection solutions/cartridges |
Lonza Bioscience |
ISO 13485; GMP-certified sites for solutions; weldable for closed systems. |
High-efficiency electroporation for hard-to-transfect cells in therapy production. |
Material specs; user responsibility for reg compliance. |
|
Raw materials (media components, enzymes, nucleotides for vector prod.) |
Roche CustomBiotech |
Precisely defined; scalable; high-quality; full traceability; regulatory compliance. |
Supports efficiency from cell isolation to QC release in viral vector processes. |
State-of-the-art biotech facility docs; pharma/diagnostics expertise. |
|
All listed products (bioprocess aids) |
Roche CustomBiotech |
Tailored to unique CGT processes; quality-controlled. |
Ensures consistency in vector production for ex vivo cell modification. |
Product portfolio specs; contact for customization. |
Report Segmentation
The market is primarily segmented based on product, end-user, and region.
|
By Product |
By End-User |
By Region |
|
|
|
To Understand the Scope of this Report: Speak to Analyst
By Product Analysis
Cell Culture Media segment is expected to witness highest growth during forecast period
The cell culture media segment is projected to grow at a CAGR during the projected period in the cell therapy human raw materials market. This is propelled by various significant factors. Cell culture media provides a controlled and optimized environment with essential nutrients, growth factors, and other critical components necessary for the survival & proliferation of cells. This controlled environment allows researchers and manufacturers to optimize conditions, ensuring the reproducibility & scalability of cell cultures for therapeutic applications. In addition, numerous companies are making substantial investments in the development of cell culture media to enhance the effectiveness of the cell therapy process.
By End-User Analysis
Biopharmaceutical and pharmaceutical companies segment is expected to dominate the cell therapy human raw materials market during forecast period
In 2025, the cell therapy human raw materials market share was predominantly influenced by biopharmaceutical & pharmaceutical companies, commanding a significant market share. The escalating demand for personalized medicine and regenerative treatments has propelled these companies to focus on developing and commercializing cutting-edge cell therapies. Advancements in technology and a supportive regulatory environment have further incentivized biopharmaceutical and pharmaceutical firms to expand their presence in this burgeoning market. The need for high-quality human raw materials, devoid of animal-origin components, is driving strategic partnerships, acquisitions, and intensified research efforts. This dynamic landscape underscores the pivotal role these companies play in shaping the future of cell therapy and underscores the potential for sustained growth in the industry.
Regional Insights
North America region dominated the global cell therapy human raw materials market in 2025
North America dominated the global cell therapy human raw materials market in 2025 and is expected to continue to do so. The prevailing influence of the region can be credited to favorable government regulations, a rising need for groundbreaking therapeutics, and concerted endeavors by major market entities to enhance cell-based therapy products addressing various chronic disorders. Additionally, North America hosts numerous prominent companies engaged in the production and commercialization of cell-based products, creating a surge in demand for cell therapy human raw materials and thereby propelling market expansion. Furthermore, collaborative initiatives between key industry players and smaller biotechnology firms heighten competition, as companies strive to fortify their positions in the dynamically evolving cell therapy market.
In the meanwhile, the Asia Pacific region is poised to experience the most rapid growth in the market. This surge is attributed to a heightened emphasis on biotechnology and a growing demand for advanced cell therapies. The market's expansion is further propelled by increased research and development activities, strategic collaborations, and the establishment of manufacturing facilities by major industry players.
Key Market Players & Competitive Insights
The cell therapy human raw materials market is fragmented and is anticipated to witness competition due to several players' presence. Major service providers in the market are constantly upgrading their technologies to stay ahead of the competition and to ensure efficiency, integrity, and safety. These players focus on partnership, product upgrades, and collaboration to gain a competitive edge over their peers and capture a significant cell therapy human raw materials market share.
Some of the major players operating in the global market include:
- Bio-Techne Corporation
- Catalent, Inc.
- CellGenix GmbH
- Corning Incorporated
- Lonza Group Ltd.
- Merck KGaA
- Miltenyi Biotec
- Pall Corporation (Danaher Corporation)
- PeproTech, Inc.
- STEMCELL Technologies Inc.
- Takara Bio Inc.
- Thermo Fisher Scientific Inc.
- WuXi AppTec
- ZenBio, Inc.
- RoosterBio Inc.
Recent Developments
- In January 2024, Solvias, a leading global provider of Chemistry, Manufacturing, and Control (CMC) analytics, disclosed its commitment to conduct analytical release testing services for the groundbreaking CRISPR/Cas9 genome-edited cell therapy. Under a long-term agreement with Vertex Pharmaceuticals, Solvias will contribute its expertise to CASGEVY, the world's first FDA-approved treatment for sickle cell disease (SCD) in patients aged 12 and above experiencing recurrent vaso-occlusive crises. This collaboration underscores Solvias' role in supporting the advancement and quality control of cutting-edge therapies in the field of genetic medicine.
- In June 2023, StemCyte revealed the formalization of a collaborative partnership with a prominent U.S. cell therapy enterprise engaged in the creation of allogeneic genetically modified CAR-NK cells derived from umbilical cord blood. Under the agreement, StemCyte commits to providing essential cellular raw materials crucial for the global advancement of allogeneic cell therapy product development.
Report Coverage
The cell therapy human raw materials market report emphasizes on key regions across the globe to provide better understanding of the product to the users. Also, the report provides market insights into recent developments, trends and analyzes the technologies that are gaining traction around the globe. Furthermore, the report covers in-depth qualitative analysis pertaining to various paradigm shifts associated with the transformation of these solutions.
The report provides detailed analysis of the cell therapy human raw materials market size while focusing on various key aspects such as competitive analysis, product, end-user, and their futuristic growth opportunities.
Cell Therapy Human Raw Materials Market Report Scope
|
Report Attributes |
Details |
|
Market size Value in 2025 |
USD 4.43 billion |
| Market size Value in 2026 | USD 5.43 billion |
|
Revenue Forecast in 2034 |
USD 28.35 billion |
|
CAGR |
22.94% from 2026 – 2034 |
|
Base Year |
2025 |
|
Historical Data |
2021 – 2024 |
|
Forecast Period |
2026 – 2034 |
|
Quantitative Units |
Revenue in USD billion and CAGR from 2026 to 2034 |
|
Segments Covered |
|
|
Regional Scope |
|
|
Competitive Landscape |
|
|
Report Format |
|
|
Customization |
Report customization as per your requirements with respect to countries, region and segmentation. |
FAQ's
The global cell therapy human raw materials market size is expected to reach USD 28.35 billion by 2034.
Corning Incorporated, Lonza Group Ltd., Merck KGaA, Miltenyi Biotec, Pall Corporation are the top market players in the market.
North America region contribute notably towards the global Cell Therapy Human Raw Materials Market.
Cell Therapy Human Raw Materials Market is exhibiting the CAGR of 22.94% during the forecast period.
The Cell Therapy Human Raw Materials Market report covering key segments are product, end-user, and region
Research Methodology
A robust system of research, verification, and forecasting designed to ensure reliable and actionable market insights.
Polaris Market Research uses a clear and structured approach to deliver insights that clients can rely on. The process combines detailed primary and secondary research, including direct communication with industry experts. The detailed information helps build a complete picture of market trends and developments. Secondary data is gathered from credible sources such as industry reports, company filings, government source links, and trusted organization databases. It is then cross-checked through discussions with key stakeholders across the value chain. Market size and forecasts are developed using both bottom-up and top-down methods to ensure accuracy and consistency in the final results.
Project Setup
Step 1 & 2:
- We start every project by clearly understanding the client’s objective or goal, then defining the market scope, and aligning regions, segments, and timelines.
- Once the foundation is set, we collect data from all-around of sources, including company reports, government databases, and paid industry platforms.
- Our research is based on secondary data, which helps us build a strong understanding of the market across regions and industries. Then we validate this information through primary research by speaking directly with industry experts, companies, and stakeholders.
- By combining secondary and primary research, we ensure that our market insights are accurate, practical, and closely aligned with real market conditions.
Data Collection
We gather information from both public and verified sources:
Data Structuring
Step 3:
- All collected data is organized into a consistent format to ensure accurate analysis. Since inputs come from multiple sources, they are standardized and aligned before use.
- The data is segmented by product, application, and region, and mapped across a defined historical period (2020–2024). All values are converted into common units (USD Mn/Bn), and volume and pricing are aligned where required to estimate revenue.
- Any overlaps or inconsistencies are reviewed and adjusted to maintain accuracy (<5% variance threshold).
- The result is a structured dataset that allows for clear comparison across regions and supports reliable analysis and forecasting.
Structured Market Dataset, USD Mn/Bn
4. Data Structuring
Step 4: TOP-DOWN APPROACH
- We start with the overall market size at a global or macro level.
- The market is then narrowed down based on scope and industry relevance.
- We apply penetration rates and split the data by region and segment.
- This helps us estimate the market size for specific segments.
- The numbers are validated through cross-checks to ensure accuracy.
Step 5: BOTTOM-UP APPROACH
- We begin by analyzing data from leading companies in the market.
- Revenue data is collected and mapped across different segments.
- The data is then aggregated to estimate the total market size.
- To fill in any gaps, adjustments are made based on industry standards.
- Validation checks make sure that the results are correct.
5. Data Structuring
Step 6:
At Polaris Market Research, we employ a methodical forecasting strategy. This approach blends the analysis of historical data with real-time market validation. To forecast future trends with precision, we examine past patterns, pricing fluctuations, and the interplay of supply and demand. To ensure our conclusions reflect the present market landscape, we actively seek input from industry experts and key stakeholders.
To refine our predictions, we carefully consider critical elements such as market drivers and restraints, fluctuations in raw material costs, emerging technologies, and the production capabilities of various regions. Furthermore, we assess regulatory frameworks and potential policy shifts to gauge their potential impact on market expansion.
All this information is synthesized to generate precise forecasts for each segment and region. These forecasts illuminate the current state of the market and highlight forthcoming opportunities.
6. Data Structuring
Step 7:
In the final stage, we validate all our estimates using a triangulation method, where data is cross-checked from multiple reliable sources, like company data, primary interviews, and secondary research. This helps us make sure that our numbers are correct and fit with the rest of the market.
This process involves verifying data consistency across various segments and geographic areas. It also requires comparing historical trends with the assumptions support the forecast. Any discrepancies involve adjustments to ensure everything remains aligned and dependable.
Once the data is finalized, we prepare the final outputs, including market size estimates, segment-wise breakdowns, and growth metrics. These are delivered in structured formats such as tables, charts, and data files for easy analysis and use.
We collaborate closely with clients, ensuring the final products align with their requirements. This includes offering tailored adjustments, supplementary data analyses, and continuous assistance. Furthermore, we monitor market trends post-delivery, providing updates and refinements to maintain the insights' relevance as time passes.
Post-delivery, we continue to monitor market shifts, offering updates and adjustments to ensure the insights remain relevant over time.
Triangulation Framework
- Company-level data
- Primary inputs from industry participants
- Secondary benchmarks and published data
- Variance maintained within ±5-10%
- Adjustments applied to align estimates
- Segment values validated against overall market structure
Data Consistency & Integrity
- Segment totals validated to 100%
- Regional estimates aligned with global market size
- Historical trends compared against forecast outputs
- Assumptions reviewed for cross-segment and regional alignment
Final Outputs
- Market size estimates (USD Mn/Bn)
- Segment-wise distribution (%)
- Growth metrics (CAGR %)
- Structured tables and charts
- Segment-level datasets
- Excel-based data files for further analysis
Client Alignment & Support
- Deliverables are aligned with defined client requirements and scope
- Custom data cuts and segment splits are incorporated as required
- Post-delivery queries are addressed through analyst interactions
- Additional clarifications and data support are provided upon request
Client Continuity & Updates
- Market developments are tracked post-delivery to capture changes in key trends
- Updated data and revisions are provided based on new market inputs
- Additional refinements and data cuts are shared as required
- Continued analyst engagement supports evolving client requirements
