What is Ophthalmic Eye Dropper Market Size?
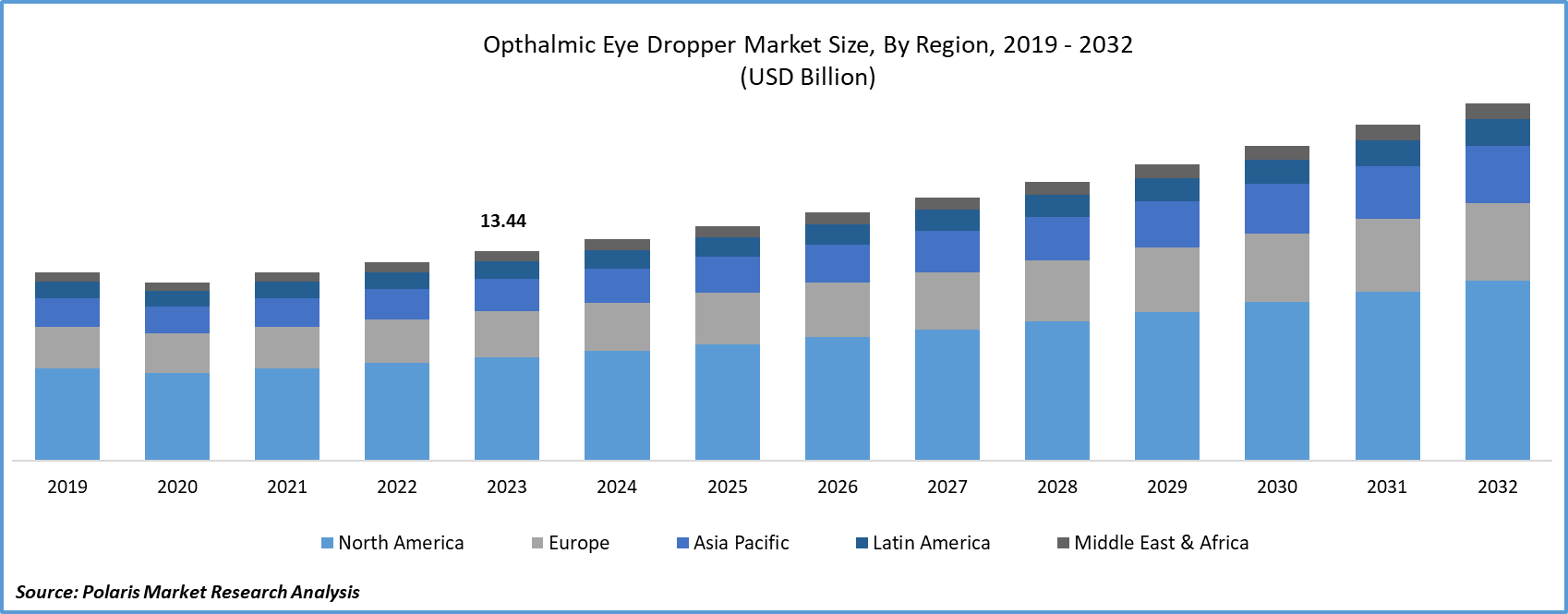
Ophthalmic Eye Dropper Market size was valued at USD 14.99 billion in 2025 and is anticipated to exhibit a CAGR of 6.0% during the forecast period. The growth is driven by rise in the prevalence of eye disease, improvement in the fast FDA approvals, and technological advancement.
Market Statistics
- 2025 Market Size: USD 14.99 Billion
- 2034 Projected Market Size: USD 25.32 Billion
- CAGR (2026-2034): 6.0%
- Largest Market: North America
Market Overview
The growth of the ophthalmic eye dropper market report is propelled by heightened eye treatment research and development investment, resulting in the development of more effective and easily adaptable tailored to diverse eye-related conditions. Moreover, the increasing global aging population contributes to a higher incidence of eye disorders like cataracts, and glaucoma, consequently amplifying the ophthalmic eye dropper’s demand.
The prevalence of eye infections is anticipated to rise globally due to the emergence of new pathogens, an increase in inadequate practice of eye hygiene, and postoperative eye infections. Common eye conditions, such as allergies, glaucoma, cataracts, and dry eyes, are becoming more widespread, leading to an increased demand for eye drops in eye care. An elderly global population significantly drives the escalating prevalence of these conditions. For instance, according to the Ministry of Health Report 2023, Glaucoma stands as the leading cause of blindness worldwide, as it is estimated that 60 million cases globally are suspected.
The growing recognition of the significance of maintaining eye health and regular eye check-ups has generated an upsurge in the demand for ophthalmic eye drops. This burgeoning awareness has resulted in an increased need for eye drops. Individuals now possess a better understanding of the early signs of eye problems, prompting them to seek assistance and treatment at an earlier stage. This heightened awareness not only enables individuals to receive timely treatment but also creates expanded opportunities for eye care professionals and manufacturers of eye care products, including eye drops. For instance, Johnson & Johnson initiated the "Vision Made Possible" program in October 2023 with the aim of educating people about various aspects of eye health, including presbyopia, childhood myopia, and cataracts. These initiatives signify a growing emphasis on taking proactive measures for better eye health, presenting increased prospects for enhancing eye health through readily available treatments like eye drops.
To Understand More About this Research: Request a Free Sample Report
The Ophthalmic Eye Dropper Market report details key market dynamics to help industry players align their business strategies with current and future trends. It examines technological advances and breakthroughs in the industry and their impact on the market presence. Furthermore, a detailed regional analysis of the industry at the local, national, and global levels has been provided.
Ophthalmic Eye Dropper Market Dynamics
Market Drivers
Prevalence of Eye Conditions bolstering the growth of the Ophthalmic Eye Dropper market industry outlook.
The prevalence of eye conditions, including common issues such as glaucoma, dry eyes, and allergies, is on the rise globally. As more individuals are experiencing these eye-related health issues, there is a growing demand for effective solutions to manage and alleviate symptoms. Ophthalmic eye droppers play a crucial role in addressing these conditions, providing a targeted and convenient method for delivering medications and therapeutic formulations directly to the eyes.
With conditions like glaucoma, which is characterized by increased intraocular pressure and dry eyes, where there is insufficient tear production or poor tear quality, regular and precise administration of medications becomes essential. Ophthalmic eye droppers offer a controlled and accurate way to deliver medications to the eyes, facilitating the management of these conditions and providing relief to individuals affected by them.
The increasing prevalence of these eye conditions has led to higher adoption of eye care regimens that include the use of ophthalmic eye droppers. Patients, in collaboration with healthcare professionals, incorporate these droppers into their routine to ensure consistent and effective treatment. As a result, the demand for ophthalmic eye droppers has been steadily increasing, driving overall market growth.
Market Restraints
Time-Consuming Approval Processes are likely to hamper the growth of the market.
The extended approval procedures for ophthalmic medications and their delivery systems present a significant hurdle within the ophthalmic eye dropper market size. Regulatory bodies, including health agencies and pharmaceutical authorities, enforce stringent guidelines to ensure the safety, effectiveness, and quality of medical products, such as ophthalmic solutions, and their associated delivery tools, such as eye droppers. Thorough evaluations, spanning preclinical studies, clinical trials, and meticulous safety and efficacy assessments, precede the market introduction of new ophthalmic products. Manufacturers are obliged to adhere to Good Manufacturing Practices (GMP) to guarantee consistent product quality.
This complex approval process involves rigorous testing, meticulous documentation, and adherence to diverse regulatory standards, resulting in a prolonged timeline from initial product development to market availability. Delays may arise at various stages, affecting design, formulation, clinical trials, and regulatory submissions.
Delayed approvals not only hinder a company's prompt response to market needs but also impede the incorporation of new technologies, restricting advancements in ophthalmic medications and delivery systems. In essence, the time-consuming nature of approval processes acts as a substantial constraint, requiring manufacturers to strategically allocate resources and navigate regulatory complexities while endeavoring to introduce cutting-edge ophthalmic products to the market.
Key Trends in Ophthalmic Eye Droppers
|
Trend |
Description |
Technical / application focus |
Examples from Practice |
|
Preservative‑free multidose squeeze dispensers |
Purely mechanical systems with spring‑loaded tip seals and sterile filter membranes prevent backflow and air contamination. |
OTC/prescription eye drops (dry eye, glaucoma); 250+ doses from 10 mL, sterile for 3 months; no preservatives needed. |
Aptar OSD launched for Restasis MultiDose (US); tip seal closes until actuation, filter vents sterile air; low force for all ages. |
|
Metered pump droppers (3K/180°) |
Pump mechanisms deliver one precise drop per stroke, independent of bottle volume or angle. |
5–10 mL bottles; viscosities up to 50 cP; CE‑marked/FDA IND for preservative‑free multidose. |
Aero Pump 3K/180° ensures reproducible dosing; side actuation ergonomic; stable force over lifecycle. |
|
Closing tip multidose systems |
Self‑closing tip orifice and one‑way valve maintain sterility without preservatives. |
Multi‑dose for glaucoma/dry eye; reproducible drop size; prevents microbial ingress. |
Nemera Novelia proven for duration of treatment; mechanical only, no chemical preservatives. |
|
Controlled drop size tips |
Engineered tips dispense 40 µL ±5 µL drops only on squeeze, avoiding accidental dispensing. |
Sterile dispensing for compounded ophthalmic meds; 3–15 mL sizes; USP <788> particulates/endotoxins tested. |
MPS Pharma dropper requires squeeze for drop; consistent size tested with water/viscous fluids. |
Report Segmentation
The market is primarily segmented based on drug type, type, treatment type, and region.
|
By Drug Type |
By Type |
By Treatment Type |
By Region |
|
|
|
|
To Understand the Scope of this Report: Speak to Analyst
Ophthalmic Eye Dropper Market Segmental Analysis
By Drug Type Analysis
In 2025, the Prescription drugs segment holds the largest share of revenue. The heightened prevalence of eye disorders, which require special prescription treatments, has generated an increased demand for eye droppers through drug administrations. Ailments like infections, dry eye ailment, and glaucoma necessitate accurately formulated medicines, which are exclusively available through prescription. Advancements in research in the pharmaceutical industry have resulted in the development of efficient and precise eye issues prescription drugs, contributing to the heightened demand within this segment. For instance, in October 2021, the launch of prescription eye drops, exemplified by FDA-approved Vuity Allergan for treating presbyopia, has been influencing the growth of the market segment.
The over-the-counter (OTC) drugs segment is experiencing profitable growth as a result of a growing consumer inclination towards self-care. The ease of access to common eye conditions medications such as redness, minor irritations, or allergies drives the use of OTC eye droppers. Furthermore, the advancing manufacturing processes and formulations have enabled the creation of OTC eye drop solutions that alleviate various eye disorders without the need for a prescription.
By Treatment Type Analysis
In the projected timeframe, the segment related to dry eye emerged as the dominant market player, attributed to the prolonged use of environmental factors, digital devices, and an elderly population, heightening the demand for targeted treatments administered through eye droppers. For instance, in January 2023, According to Environmental Research and Public Health, Dry Eye Disease (DED) ranks among the most widespread eye conditions affecting millions globally. The prevalence of this condition varies across the globe, ranging from 5% to 50% based on different locations. This growing prevalence emphasizes the increased demand for treatments administered through eye droppers to tackle the challenges presented by dry eye syndrome effectively.
Ophthalmic Eye Dropper Market Regional Insights
The North American region dominated the global market with the largest market share in 2025
This is attributed to substantial healthcare spending, enabling broad accessibility to recently developed eye care products and treatments such as eye droppers. Furthermore, the rising incidence of eye infections and allergies and an expanding elderly population have generated increased demand for eye solutions, fostering market expansion in North America. Additionally, ongoing technological advancements, investing is research and development, have led to the introduction of innovative eye care medications and products. The ophthalmic eye dropper companies have played a role in market growth by introducing innovation and advancement in eye droppers.
The United States dominated the North American market, primarily owing to its comprehensive healthcare system and extensive availability of new eye care products and treatments, including eye droppers. Furthermore, the U.S. continues to lead in research and innovation within the healthcare sector, consistently introducing advancement in formulations and technologies in the field of ophthalmology.
Asia Pacific is poised to achieve the fastest growth. The increasing population and growing concerns about eye diseases and health are significant factors propelling the use and demand for eye droppers and solutions for eye care. Furthermore, shifts in lifestyle, escalating eye diseases among the elderly population, and heightened exposure to digital devices help in increasing demand for ophthalmic medications.
Competitive Landscape
The Ophthalmic Eye Dropper market share is fragmented and is anticipated to witness competition due to several players' presence. Major service providers in the market are constantly upgrading their technologies to stay ahead of the competition and to ensure efficiency, integrity, and safety. These players focus on partnership, product upgrades, and collaboration to gain a competitive edge over their peers and capture a significant market share.
Some of the major players operating in the global market include:
- AbbVie Inc.
- Alcon
- AptarGroup, Inc.
- Bausch & Lomb Incorporated
- Novartis AG
- Pfizer
- Silgan Dispensing Systems
Recent Developments
- July 2025, LENZ Therapeutics announced the U.S. FDA approval and launch of VIZZ™, the first aceclidine-based once-daily eye drop for presbyopia, enabling adults to improve near vision for up to 10 hours and marking a major advancement in age-related vision treatment
- September 2023, RYZUMVlTM (Phentolamine Ophthalmic Solution) 0.75% Eye Drops received FDA approval. These eye drops are formulated to manage pharmacologically induced mydriasis caused by parasympatholytic drugs (like Tropicamide) or adrenergic agonists (such as Phenylephrine).
- May 2023, Bausch + Lomb Corporation obtained FDA approval for MIEBO (perfluorohexyloctane ophthalmic solution, previously known as NOV03). This achievement marks the first FDA-approved medication in the U.S. specifically designed to address the symptoms and signs of dry eye disease (DED).
- April 2022, Sandoz, a subsidiary of Novartis AG, launched a new generic brimonidine tartrate/timolol maleate eye drop in the United States. This expansion of treatment options for ocular hypertension patients strengthens its position in the ophthalmic market.
Report Coverage
The Ophthalmic Eye Dropper market industry trends report emphasizes key regions across the globe to provide a better understanding of the product to the users. Also, the report provides market insights into recent developments and trends and analyzes the technologies that are gaining traction around the globe. Furthermore, the report covers an in-depth qualitative analysis pertaining to various paradigm shifts associated with the transformation of these solutions.
The report provides a detailed analysis of the market while focusing on various key aspects such as competitive analysis, drug type, type, treatment type, and their futuristic growth opportunities.
Ophthalmic Eye Dropper Market Report Scope
|
Report Attributes |
Details |
|
Market size in 2025 |
USD 14.99 billion |
| Market size in 2026 | USD 15.83 billion |
|
Revenue forecast in 2034 |
USD 25.32 billion |
|
CAGR |
6.0% from 2026 – 2034 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2034 |
|
Quantitative units |
Revenue in USD billion and CAGR from 2026 to 2034 |
|
Segments covered |
By Drug type, By Type, By Treatment type, By Region |
|
Regional scope |
North America, Europe, Asia Pacific, Latin America; Middle East & Africa |
|
Customization |
Report customization as per your requirements with respect to countries, regions, and segmentation. |
FAQ's
The Ophthalmic Eye Dropper Market Market report covering key segments are drug type, type, treatment type, and region.
Ophthalmic Eye Dropper Market Size Worth $25.32 Billion By 2034
Ophthalmic Eye Dropper Market exhibiting a CAGR of 6.0% during the forecast period
North American is leading the global market
key driving factors in Ophthalmic Eye Dropper Market are Rising customized treatments
Research Methodology
A robust system of research, verification, and forecasting designed to ensure reliable and actionable market insights.
Polaris Market Research uses a clear and structured approach to deliver insights that clients can rely on. The process combines detailed primary and secondary research, including direct communication with industry experts. The detailed information helps build a complete picture of market trends and developments. Secondary data is gathered from credible sources such as industry reports, company filings, government source links, and trusted organization databases. It is then cross-checked through discussions with key stakeholders across the value chain. Market size and forecasts are developed using both bottom-up and top-down methods to ensure accuracy and consistency in the final results.
Project Setup
Step 1 & 2:
- We start every project by clearly understanding the client’s objective or goal, then defining the market scope, and aligning regions, segments, and timelines.
- Once the foundation is set, we collect data from all-around of sources, including company reports, government databases, and paid industry platforms.
- Our research is based on secondary data, which helps us build a strong understanding of the market across regions and industries. Then we validate this information through primary research by speaking directly with industry experts, companies, and stakeholders.
- By combining secondary and primary research, we ensure that our market insights are accurate, practical, and closely aligned with real market conditions.
Data Collection
We gather information from both public and verified sources:
Data Structuring
Step 3:
- All collected data is organized into a consistent format to ensure accurate analysis. Since inputs come from multiple sources, they are standardized and aligned before use.
- The data is segmented by product, application, and region, and mapped across a defined historical period (2020–2024). All values are converted into common units (USD Mn/Bn), and volume and pricing are aligned where required to estimate revenue.
- Any overlaps or inconsistencies are reviewed and adjusted to maintain accuracy (<5% variance threshold).
- The result is a structured dataset that allows for clear comparison across regions and supports reliable analysis and forecasting.
Structured Market Dataset, USD Mn/Bn
4. Data Structuring
Step 4: TOP-DOWN APPROACH
- We start with the overall market size at a global or macro level.
- The market is then narrowed down based on scope and industry relevance.
- We apply penetration rates and split the data by region and segment.
- This helps us estimate the market size for specific segments.
- The numbers are validated through cross-checks to ensure accuracy.
Step 5: BOTTOM-UP APPROACH
- We begin by analyzing data from leading companies in the market.
- Revenue data is collected and mapped across different segments.
- The data is then aggregated to estimate the total market size.
- To fill in any gaps, adjustments are made based on industry standards.
- Validation checks make sure that the results are correct.
5. Data Structuring
Step 6:
At Polaris Market Research, we employ a methodical forecasting strategy. This approach blends the analysis of historical data with real-time market validation. To forecast future trends with precision, we examine past patterns, pricing fluctuations, and the interplay of supply and demand. To ensure our conclusions reflect the present market landscape, we actively seek input from industry experts and key stakeholders.
To refine our predictions, we carefully consider critical elements such as market drivers and restraints, fluctuations in raw material costs, emerging technologies, and the production capabilities of various regions. Furthermore, we assess regulatory frameworks and potential policy shifts to gauge their potential impact on market expansion.
All this information is synthesized to generate precise forecasts for each segment and region. These forecasts illuminate the current state of the market and highlight forthcoming opportunities.
6. Data Structuring
Step 7:
In the final stage, we validate all our estimates using a triangulation method, where data is cross-checked from multiple reliable sources, like company data, primary interviews, and secondary research. This helps us make sure that our numbers are correct and fit with the rest of the market.
This process involves verifying data consistency across various segments and geographic areas. It also requires comparing historical trends with the assumptions support the forecast. Any discrepancies involve adjustments to ensure everything remains aligned and dependable.
Once the data is finalized, we prepare the final outputs, including market size estimates, segment-wise breakdowns, and growth metrics. These are delivered in structured formats such as tables, charts, and data files for easy analysis and use.
We collaborate closely with clients, ensuring the final products align with their requirements. This includes offering tailored adjustments, supplementary data analyses, and continuous assistance. Furthermore, we monitor market trends post-delivery, providing updates and refinements to maintain the insights' relevance as time passes.
Post-delivery, we continue to monitor market shifts, offering updates and adjustments to ensure the insights remain relevant over time.
Triangulation Framework
- Company-level data
- Primary inputs from industry participants
- Secondary benchmarks and published data
- Variance maintained within ±5-10%
- Adjustments applied to align estimates
- Segment values validated against overall market structure
Data Consistency & Integrity
- Segment totals validated to 100%
- Regional estimates aligned with global market size
- Historical trends compared against forecast outputs
- Assumptions reviewed for cross-segment and regional alignment
Final Outputs
- Market size estimates (USD Mn/Bn)
- Segment-wise distribution (%)
- Growth metrics (CAGR %)
- Structured tables and charts
- Segment-level datasets
- Excel-based data files for further analysis
Client Alignment & Support
- Deliverables are aligned with defined client requirements and scope
- Custom data cuts and segment splits are incorporated as required
- Post-delivery queries are addressed through analyst interactions
- Additional clarifications and data support are provided upon request
Client Continuity & Updates
- Market developments are tracked post-delivery to capture changes in key trends
- Updated data and revisions are provided based on new market inputs
- Additional refinements and data cuts are shared as required
- Continued analyst engagement supports evolving client requirements
