Market Overview
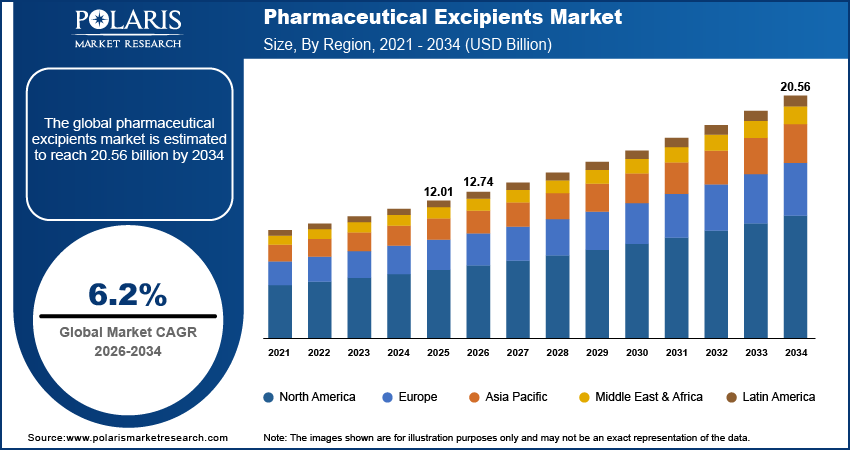
The pharmaceutical excipients market size was valued at USD 12.01 billion in 2025, growing at a CAGR of 6.2% during 2026 to 2034. The expanding pharmaceutical industry and increased demand for functional excipients that enhance bioavailability are among the key factors driving market growth.
Pharmaceutical excipients are gaining prominence as pharmaceutical companies seek ways to improve drug performance. Excipients are used to improve solubility, stability, taste, and controlled release of drugs, making them more effective and easier to take. Excipients are used in tablets as well as injectable drugs. They are also significant in improving production efficiency and facilitating mass production.
Near-term (12-24 months) forecast: The growing capacity additions in the pharmaceutical industry and the rise in generic production are anticipated to fuel demand for high-quality excipients. The market also benefits from the continued success of biologic drugs. This includes the use of co-processed excipients and specialty grades of excipients that enable advanced drug delivery systems.
Key Insights
- The organic chemicals segment accounts for a larger market share. The versatility and compatibility of organic chemicals drive their leading market share.
- Organic excipients, such as cellulose derivatives, carbohydrates, and proteins, remain at the forefront of the market owing to their broad compatibility with tablet dosage forms. On the other hand, parenteral excipients are emerging as a popular choice with the growing number of injectable drugs and biologics in the pipeline.
- The tablets formulation segment is projected to register significant growth. The stability and cost-effectiveness of tablets make them a preferred dosage form.
- The Europe pharmaceutical excipients market accounts for a larger market share. The regional market dominance is attributed to its stringent regulatory framework and well-established pharmaceutical sector.
- The Asia Pacific pharmaceutical excipients market is registering the fastest growth. The introduction of favorable government policies and rising healthcare expenditures are a few of the key factors driving the regional market growth.
Industry Dynamics
- Rising investments by pharmaceutical companies in the expansion of their manufacturing facilities is fueling the demand for pharmaceutical excipients in drug manufacturing, driving market growth.
- Increased efforts for reforming clinical trial regulations are another key factor fueling market expansion.
- Advancements in drug delivery formulations are expected to provide several market opportunities.
- Regulatory compliance and stringent quality standards may hinder market growth.
Market Statistics
2025 Market Size: USD 12.01 billion
2034 Projected Market Size: USD 20.56 billion
CAGR (2026-2034): 6.2%
Europe: Largest Market in 2025
To Understand More About this Research:Request a Free Sample Report
The pharmaceutical excipients market comprises the various excipients used in drug formulations to improve drug stability, bioavailability, and patient acceptability. Excipients serve as carriers, fillers, binders, disintegrating agents, preservatives, and coating agents. Excipients are used to ensure the right delivery and performance of active pharmaceutical ingredients (APIs). The market is fueled by the rise in the pharmaceutical industry and technological developments in pharmaceutical formulations. The market is further fueled by rising demand for functional excipients that improve drug solubility and bioavailability. Additionally, the rising prevalence of chronic diseases and the expansion of the generic and biologic markets are driving growth in the pharmaceutical excipients market.
|
Function |
Example Excipients |
Best-Fit Dosage Forms |
|
Binders (Pharmaceutical Binders) |
Starch, cellulose derivatives |
Tablets, capsules |
|
Disintegrants (Disintegrants in Pharmaceuticals) |
Croscarmellose sodium, sodium starch glycolate |
Tablets that need to break apart quickly after swallowing |
|
Coating Agents (Coating Agents Excipients) |
Film-forming polymers, sugar coatings |
Tablets, modified-release tablets |
|
Fillers/Diluents |
Lactose, microcrystalline cellulose |
Tablets, capsules |
|
Preservatives |
Parabens, benzyl alcohol |
Liquid syrups, injectables |
|
Lubricants/Glidants |
Magnesium stearate, colloidal silica |
Tablets, capsules |
The key factors driving the pharmaceutical excipients market are the growing use of advanced drug delivery systems, the rising demand for oral and parenteral dosage forms, and the strict regulatory standards that encourage the use of high-quality excipients. The rising demand for multifunctional excipients and co-processed excipients to optimize drug formulation processes is also driving growth in the pharmaceutical excipients market. Multifunctional excipients and co-processed excipients simplify manufacturing by enabling multiple functionalities in a single excipient. This results in fewer processing steps, improved powder flow properties, and easier use in high-speed tablet presses. This, in turn, enables the manufacturer to have improved production efficiency and quality. In addition, the rising trend of biopharmaceuticals and personalized medicine is fueling the use of advanced excipients with improved performance properties.
Market Dynamics
Rising Investments in Pharmaceutical Manufacturing
Companies across the globe are investing heavily in expanding manufacturing facilities. For example, in February 2025, Eli Lilly made a USD 27 billion investment to create four manufacturing mega-sites in the US. The heavy investment in expanding the manufacturing facility is thus increasing demand for pharmaceutical excipients used in the manufacture of drugs. Furthermore, government initiatives are expected to boost the demand for excipients sourced from within the country, as pharmaceutical companies look to comply with the new regulations and take advantage of government incentives, thus fueling the growth of the pharmaceutical excipients market.
The expanding pharmaceutical manufacturing industry requires excipients that meet quality standards during high-speed production. This is especially true for tablet excipients such as binders, fillers and diluents, and coating agents that allow smooth large-scale tablet production. The injectable formulation requires parenteral excipients to fulfill two essential requirements. These include achieving strict purity standards and delivering comprehensive documentation.
Regulatory Reforms Enhancing Clinical Trial Processes
The efforts for the reform of clinical trial regulations are propelling the demand for the pharmaceutical excipients market. The improvement in regulatory reforms is encouraging the simplification of regulatory procedures and the provision of incentives to increase the number of clinical trials. This is expected to boost pharmaceutical research and development, thereby increasing demand for specialized excipients used in different drug formulations.
The demand for specialty excipients is increasing due to faster clinical trial development, especially for modified-release dosage forms, pediatric formulations, and biologics. These dosage forms need excipients that are stable and compatible with the sensitive ingredients. The demand for stability and compatibility is more challenging than for conventional small-molecule tablets.
Regulatory & Quality Compliance Landscape
The pharmaceutical excipients industry considers regulatory compliance a core aspect of purchasing and formulation. The industry has witnessed an increasing demand for pharmacopeial acceptance and compatibility with sound quality management systems. There is a significant focus on documentation readiness, including specifications, certificates of analysis (CoA), and change control processes, as well as the ability to audit suppliers. This enables the formulation of risk-free products and prevents regulatory approval delays, especially for sterile and injectable products.
What Buyers Check (Excipient Documentation & Compliance)
USP-NF / Ph. Eur. Harmonization: Documentation of the excipient’s conformance to accepted quality standards (USP-NF excipients, Ph. Eur. excipients).
Availability of DMF / CEP (where applicable): Supporting documentation that facilitates faster approval of products.
Impurity controls: Clear information demonstrating that the unwanted material is controlled within safe levels.
Microbiological limits: Proof that the material is free from harmful microorganisms, especially important in sterile and injectable products.
Traceability and excipient supplier qualification: Traceability of the material to its origin, along with proper excipient supplier qualification and excipient documentation.
Supply Chain & Sourcing Analysis
The excipient supply chain involves commodity and specialty excipients such as cellulose, lactose, starch derivatives, lipids, and polymers. This supply chain is very sensitive to quality consistency, delivery times, and quality regulatory documentation. To mitigate supply chain risks, pharmaceutical companies are increasingly adopting dual sourcing and supplier qualification processes, especially for excipients used in parenteral products and biologics, which may require additional testing and regulatory approval if a supplier change is made.

Market Segment Insights
Assessment – By Product
The pharmaceutical excipients market, by product, is segmented into inorganic chemicals and organic chemicals. The organic chemicals segment holds a larger share of the pharmaceutical excipients market. This is due to their versatility and compatibility with pharmaceutical formulations. This segment comprises oleochemicals, carbohydrates, petrochemicals, and proteins. These organic chemicals are essential for improving the stability, bioavailability, and patient acceptability of pharmaceuticals. The rising demand for organic excipients is primarily due to their non-toxic nature and eco-friendliness. In addition to this, the rising preference for natural and plant-based products has increased the demand for organic excipients. The demand for organic excipients is expected to rise in the coming years due to their eco-friendliness.
Key organic excipients such as cellulose derivatives and lactose-based excipients are commonly employed in oral solid dosage forms because of their ability to provide good compressibility and smooth flow properties during manufacturing. These properties have continued to contribute to the prominent role of organic materials in tablet formulation.
Evaluation – By Formulation
The pharmaceutical excipients market, by formulation, is segmented into tablets formulation, capsules formulation, parenteral formulation, liquid formulation, and oral formulation. The tablets formulation segment is anticipated to register substantial growth during the forecast period. This is due to the popularity of tablets as a preferred dosage form owing to their convenience, stability, and cost-effectiveness. Tablets provide accurate dosing, ease of administration, and a long shelf life, which are factors contributing to their widespread use in various therapeutic classes. The rising incidence of chronic diseases and the growing need for oral solid dosage forms also support the growth of the tablets formulation segment. Furthermore, technological advancements in tablet manufacturing, such as coated and controlled-release tablets, improve patient compliance and fuel the growth of this segment.
Evaluation – By Functionality
By functionality, the market includes binders, fillers & diluents, disintegrants, coating agents, preservatives, and suspending & viscosity agents. There is growing demand for performance-enhancing agents, such as co-processed excipients that improve tablet hardness and uniformity, and specialty excipients for injectables that help maintain stability and shelf life.
Regional Insights
By region, the study provides pharmaceutical excipients market insights into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. The Europe pharmaceutical excipients market represents the largest market share. This can be attributed to the well-established pharmaceutical industry and the strict regulatory environment in the region. The region also experiences constant innovations in drug formulation technologies. The presence of large pharmaceutical companies and contract manufacturing organizations has also fueled the demand for high-quality excipients in the region. The European Medicines Agency (EMA) has also enforced strict quality standards, and this has contributed to the adoption of functional and co-processed excipients to enhance drug formulations. The increasing demand for generic and biosimilar drugs, particularly in Germany, France, and the UK, has also contributed to the growth of the market. The innovation and sustainability trend in the region has also contributed to the adoption of naturally derived excipients.
The Asia Pacific pharmaceutical excipients market is growing at a fast pace. The market is fueled by the growing pharmaceutical manufacturing industry and rising healthcare spending. The market is also fueled by the support of favorable government policies. Regions such as China, India, and Japan are major contributors to the market. China and India are large centers for pharmaceutical manufacturing and export. The favorable manufacturing environment, a strong base of generic pharmaceutical manufacturing, and increasing investments in R&D are propelling the market. Government initiatives aimed at strengthening the domestic pharmaceutical manufacturing base and expanding access to drugs are also driving the increasing demand for excipients. The increasing prevalence of chronic diseases and the adoption of innovative drug-delivery systems have also driven demand for specialty excipients.
Key Players and Competitive Insights
The pharmaceutical excipients market has several prominent companies that actively contribute to the industry. These include The Lubrizol Corporation; Archer Daniels Midland Company; DuPont de Nemours, Inc.; BASF SE; Evonik Industries AG; Ashland Global Holdings Inc.; Roquette Frères; Kerry Group plc; Croda International Plc; DFE Pharma; JRS Pharma; Colorcon, Inc.; MEGGLE GmbH & Co. KG; Wacker Chemie AG; and Associated British Foods plc. Each of these entities plays a vital role in supplying essential excipients for pharmaceutical formulations.
Competitive differentiation is being driven by high-value segments that improve manufacturing efficiency. These include parenteral-grade excipients, biologics stabilizers, taste-masking systems, and co-processed excipients. The companies are positioning themselves by providing strong regulatory documentation, application labs, and formulation support services that help pharmaceutical companies accelerate development.
Kerry Group Plc provides a broad range of taste and nutrition solutions, such as excipients that enhance the palatability of drugs. Croda International Plc offers specialized lipid excipients. Its excipients make it easier to develop advanced drug delivery systems. DFE Pharma and JRS Pharma are recognized for their expertise in excipients. They provide high-quality lactose excipients and cellulose excipients. Colorcon, Inc. specializes in film-coating technologies that improve the appearance and performance of oral dosage forms. MEGGLE GmbH & Co. KG is a company that provides lactose excipients, while Wacker Chemie AG provides cyclodextrins and other specialty excipients. Associated British Foods Plc is a company that provides a range of excipients through its subsidiary SPI Pharma, which focuses on patient-friendly drug formulations.
The Lubrizol Corporation, a subsidiary of Berkshire Hathaway, focuses on specialty chemicals. It provides solutions that improve the performance of products across industries, including pharmaceuticals. The company's knowledge of polymer technologies and excipients is very important in drug formulation and drug delivery systems.
BASF SE, a German-based company, is a leading chemical business. It has a broad portfolio of chemicals, materials, and solutions for different industries. In the pharmaceutical excipients industry, BASF provides a broad portfolio of products that help in drug formulation, stability, and bioavailability.
List of Key Companies
- Archer Daniels Midland Company
- Ashland Global Holdings Inc.
- Associated British Foods plc
- BASF SE
- Colorcon, Inc.
- Croda International Plc
- DFE Pharma
- DuPont de Nemours, Inc.
- Evonik Industries AG
- JRS Pharma
- Kerry Group plc
- MEGGLE GmbH & Co. KG
- Roquette Frères
- The Lubrizol Corporation
- Wacker Chemie AG
Pharmaceutical Excipients Industry Developments
- May 2025: Roquette revealed the completion of the acquisition of IFF Pharma Solutions. According to Roquette, the acquisition is a major step in its goal of becoming a key player in support of the global pharma markets.
- November 2024: Nagase Vitta launched an injectable-grade stabilizer, SUCROSE SG. The firm disclosed that the stabilizer is eco-friendly. It aims to enhance the stability of biopharmaceuticals.
- November 2024: The new high-performance excipients from Clariant were launched at CPHI India 2024, featuring products for sensitive APIs, parenteral formulations, and colorless applications. The company stated that the new excipients aim to improve global pharmaceutical solutions and compliance.
- October 2023: Three new excipient grades, LYCATAB CT-LM, MICROCEL 103 SD, and MICROCEL 113 SD, were launched by Roquette at CPHI Barcelona, which will improve moisture protection for moisture-sensitive pharmaceutical formulations.
Pharmaceutical Excipients Market Segmentation
By Product Outlook (Revenue – USD Billion, 2021–2034)
- Inorganic Chemicals
- Organic Chemicals
By Formulation Outlook (Revenue – USD Billion, 2021–2034)
- Tablets Formulation
- Capsules Formulation
- Parental Formulation
- Liquid Formulation
- Oral Formulation
By Functionality Outlook (Revenue – USD Billion, 2021–2034)
- Blinders
- Fillers & Diluents
- Suspending & Viscosity agents
- Preservatives
- Flavoring Agents & Sweeteners
- Coating Agents
By End Use Outlook (Revenue – USD Billion, 2021–2034)
- Pharmaceutical Companies
- Contract Manufacturing Organizations
- Research Institutions
By Regional Outlook (Revenue – USD Billion, 2021–2034)
- North America
- US
- Canada
- Europe
- Germany
- France
- UK
- Italy
- Spain
- Netherlands
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Malaysia
- South Korea
- Indonesia
- Australia
- Vietnam
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Israel
- South Africa
- Rest of Middle East & Africa
- Latin America
- Mexico
- Brazil
- Argentina
- Rest of Latin America
Pharmaceutical Excipients Market Report Scope
|
Report Attributes |
Details |
|
Market Size in 2025 |
USD 12.01 billion |
|
Market Size in 2026 |
USD 12.74 billion |
|
Revenue Forecast by 2034 |
USD 20.56 billion |
|
CAGR |
6.2% |
|
Base Year |
2025 |
|
Historical Data |
2021–2024 |
|
Forecast Period |
2026–2034 |
|
Quantitative Units |
Revenue in USD billion, and CAGR from 2026 to 2034 |
|
Report Coverage |
Revenue Forecast, Market Competitive Landscape, Growth Factors, and Trends |
|
Segments Covered |
|
|
Regional Scope |
|
|
Competitive Landscape |
Pharmaceutical Excipients Industry Trend Analysis (2025) Company profiles/industry participants profiling include company overview, financial information, product/service benchmarking, and recent developments |
|
Report Format |
PDF + Excel |
|
Customization |
Report customization as per your requirements with respect to countries, regions, and segmentation. |
How is the report valuable for an organization?
Workflow/Innovation Strategy
The pharmaceutical excipients market has been segmented into detailed segments of product, formulation, functionality, and end use. Moreover, the study provides the reader with a detailed understanding of the different segments at both the global and regional levels.
Growth/Marketing Strategy
The growth of the pharmaceutical excipients market is driven by innovation, collaboration, and geographic expansion. The companies are investing in R&D to develop multifunctional, co-processed excipients that improve the efficiency of drug formulations and patient compliance. Collaborations with pharmaceutical companies and contract research organizations (CROs) are enhancing the distribution channels and market access. Compliance with regulations and quality standards is a major priority for ensuring product acceptance in the global market. Sustainability initiatives, such as the development of plant-based and biodegradable excipients, are also gaining popularity to meet industry trends and requirements.
FAQ's
The pharmaceutical excipients market stood at USD 12.01 billion in 2025. It is projected to reach USD 20.56 billion by 2034.
The market is projected to account for a CAGR of 6.2% between 2026 and 2034.
Europe accounted for the largest market share in 2025 due to its well-established pharmaceutical industry.
A few of the key players in the market include The Lubrizol Corporation; Archer Daniels Midland Company; DuPont de Nemours, Inc.; BASF SE; Evonik Industries AG; Ashland Global Holdings Inc.; Roquette Frères; Kerry Group plc; Croda International Plc; DFE Pharma; JRS Pharma; Colorcon, Inc.; MEGGLE GmbH & Co. KG; Wacker Chemie AG; and Associated British Foods plc.
The organic chemicals segment accounted for the largest market share in 2025 due to its versatility and compatibility in drug formulation.
The tablets formulations segment is expected to witness significant growth, owing to the widespread use of tablets as a preferred dosage form.
Pharmaceutical excipients are inactive ingredients used in medicines to help with the effective delivery of drugs. These excipients support stability, texture, and shelf life.
Common types include fillers, binders, preservatives, flavoring agents, and coating materials.
Yes. Excipients have to be of high quality and safe before they can be accepted for use in medications.
The pharmaceutical excipients market is growing due to the rising production of pharmaceuticals and the growing demand for generics.
Each of these has to use different excipients to achieve the right stability and absorption.
These are excipients that have more than one function, such as a binder and a filler. Their use helps simplify manufacturing.
Research Methodology
A robust system of research, verification, and forecasting designed to ensure reliable and actionable market insights.
Polaris Market Research uses a clear and structured approach to deliver insights that clients can rely on. The process combines detailed primary and secondary research, including direct communication with industry experts. The detailed information helps build a complete picture of market trends and developments. Secondary data is gathered from credible sources such as industry reports, company filings, government source links, and trusted organization databases. It is then cross-checked through discussions with key stakeholders across the value chain. Market size and forecasts are developed using both bottom-up and top-down methods to ensure accuracy and consistency in the final results.
Project Setup
Step 1 & 2:
- We start every project by clearly understanding the client’s objective or goal, then defining the market scope, and aligning regions, segments, and timelines.
- Once the foundation is set, we collect data from all-around of sources, including company reports, government databases, and paid industry platforms.
- Our research is based on secondary data, which helps us build a strong understanding of the market across regions and industries. Then we validate this information through primary research by speaking directly with industry experts, companies, and stakeholders.
- By combining secondary and primary research, we ensure that our market insights are accurate, practical, and closely aligned with real market conditions.
Data Collection
We gather information from both public and verified sources:
Data Structuring
Step 3:
- All collected data is organized into a consistent format to ensure accurate analysis. Since inputs come from multiple sources, they are standardized and aligned before use.
- The data is segmented by product, application, and region, and mapped across a defined historical period (2020–2024). All values are converted into common units (USD Mn/Bn), and volume and pricing are aligned where required to estimate revenue.
- Any overlaps or inconsistencies are reviewed and adjusted to maintain accuracy (<5% variance threshold).
- The result is a structured dataset that allows for clear comparison across regions and supports reliable analysis and forecasting.
Structured Market Dataset, USD Mn/Bn
4. Data Structuring
Step 4: TOP-DOWN APPROACH
- We start with the overall market size at a global or macro level.
- The market is then narrowed down based on scope and industry relevance.
- We apply penetration rates and split the data by region and segment.
- This helps us estimate the market size for specific segments.
- The numbers are validated through cross-checks to ensure accuracy.
Step 5: BOTTOM-UP APPROACH
- We begin by analyzing data from leading companies in the market.
- Revenue data is collected and mapped across different segments.
- The data is then aggregated to estimate the total market size.
- To fill in any gaps, adjustments are made based on industry standards.
- Validation checks make sure that the results are correct.
5. Data Structuring
Step 6:
At Polaris Market Research, we employ a methodical forecasting strategy. This approach blends the analysis of historical data with real-time market validation. To forecast future trends with precision, we examine past patterns, pricing fluctuations, and the interplay of supply and demand. To ensure our conclusions reflect the present market landscape, we actively seek input from industry experts and key stakeholders.
To refine our predictions, we carefully consider critical elements such as market drivers and restraints, fluctuations in raw material costs, emerging technologies, and the production capabilities of various regions. Furthermore, we assess regulatory frameworks and potential policy shifts to gauge their potential impact on market expansion.
All this information is synthesized to generate precise forecasts for each segment and region. These forecasts illuminate the current state of the market and highlight forthcoming opportunities.
6. Data Structuring
Step 7:
In the final stage, we validate all our estimates using a triangulation method, where data is cross-checked from multiple reliable sources, like company data, primary interviews, and secondary research. This helps us make sure that our numbers are correct and fit with the rest of the market.
This process involves verifying data consistency across various segments and geographic areas. It also requires comparing historical trends with the assumptions support the forecast. Any discrepancies involve adjustments to ensure everything remains aligned and dependable.
Once the data is finalized, we prepare the final outputs, including market size estimates, segment-wise breakdowns, and growth metrics. These are delivered in structured formats such as tables, charts, and data files for easy analysis and use.
We collaborate closely with clients, ensuring the final products align with their requirements. This includes offering tailored adjustments, supplementary data analyses, and continuous assistance. Furthermore, we monitor market trends post-delivery, providing updates and refinements to maintain the insights' relevance as time passes.
Post-delivery, we continue to monitor market shifts, offering updates and adjustments to ensure the insights remain relevant over time.
Triangulation Framework
- Company-level data
- Primary inputs from industry participants
- Secondary benchmarks and published data
- Variance maintained within ±5-10%
- Adjustments applied to align estimates
- Segment values validated against overall market structure
Data Consistency & Integrity
- Segment totals validated to 100%
- Regional estimates aligned with global market size
- Historical trends compared against forecast outputs
- Assumptions reviewed for cross-segment and regional alignment
Final Outputs
- Market size estimates (USD Mn/Bn)
- Segment-wise distribution (%)
- Growth metrics (CAGR %)
- Structured tables and charts
- Segment-level datasets
- Excel-based data files for further analysis
Client Alignment & Support
- Deliverables are aligned with defined client requirements and scope
- Custom data cuts and segment splits are incorporated as required
- Post-delivery queries are addressed through analyst interactions
- Additional clarifications and data support are provided upon request
Client Continuity & Updates
- Market developments are tracked post-delivery to capture changes in key trends
- Updated data and revisions are provided based on new market inputs
- Additional refinements and data cuts are shared as required
- Continued analyst engagement supports evolving client requirements
